19.3
Two Photosystems Generate a Proton Gradient and NADPH in Oxygenic Photosynthesis
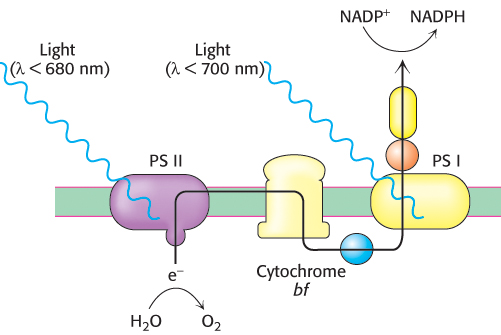
FIGURE 19.12Two photosystems. The absorption of photons by two distinct photosystems (PS I and PS II) is required for complete electron flow from water to NADP+.
Photosynthesis is more complicated in green plants than in photosynthetic bacteria. In green plants, photosynthesis depends on the interplay of two kinds of membrane-bound, light-sensitive complexes— photosystem I (PS I) and photosystem II (PS II), as shown in Figure 19.12. There are similarities in photosynthesis between green plants and photosynthetic bacteria. Both require light to energize reaction centers consisting of special pairs, called P700 for photosystem I and P680 for photosystem II, and both transfer electrons by using electron-transport chains. However, in plants, electron flow is not cyclic but progresses from photosystem II to photosystem I under most circumstances.
Photosystem I, which responds to light with wavelengths shorter than 700 nm, uses light-derived high-energy electrons to create biosynthetic reducing power in the form of NADPH, a versatile reagent for driving biosynthetic processes. The electrons for creating one molecule of NADPH are taken from two molecules of water by photosystem II, which responds to wavelengths shorter than 680 nm. A molecule of O2 is generated as a side product of the actions of photosystem II. The electrons travel from photosystem II to photosystem I through cytochrome bf, a membrane-bound complex homologous to Complex III in oxidative phosphorylation. Cytochrome bf generates a proton gradient across the thylakoid membrane that drives the formation of ATP. Thus, the two photosystems cooperate to produce NADPH and ATP.
Photosystem II transfers electrons from water to plastoquinone and generates a proton gradient
Photosystem II, an enormous transmembrane assembly of more than 20 subunits, catalyzes the light-driven transfer of electrons from water to plastoquinone. This electron acceptor closely resembles ubiquinone, a component of the mitochondrial electron-transport chain. Plastoquinone cycles between an oxidized form (Q) and a reduced form (QH2, plastoquinol). The overall reaction catalyzed by photosystem II is
The electrons in QH2 are at a higher redox potential than those in H2O. Recall that, in oxidative phosphorylation, electrons flow from ubiquinol to an acceptor, O2, which is at a lower potential. Photosystem II drives the reaction in a thermodynamically uphill direction by using the free energy of light.
This reaction is similar to one catalyzed by the bacterial system in that a quinone is converted from its oxidized into its reduced form. Photosystem II is reasonably similar to the bacterial reaction center (Figure 19.13). The core of the photosystem is formed by D1 and D2, a pair of similar 32-kDa subunits that span the thylakoid membrane. These subunits are homologous to the L and M chains of the bacterial reaction center. Unlike the bacterial system, photosystem II contains a large number of additional subunits that bind more than 30 chlorophyll molecules altogether and increase the efficiency with which light energy is absorbed and transferred to the reaction center (Section 19.5).
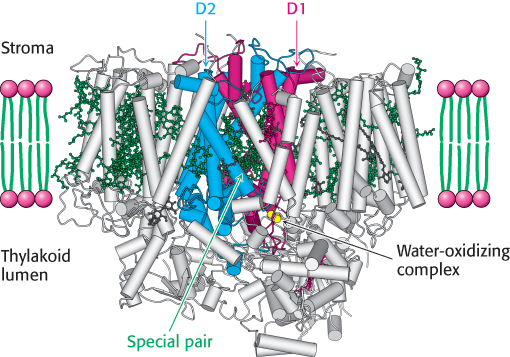
 FIGURE 19.13 The structure of photosystem II. The D1 (red) and D2 (blue) subunits and the numerous bound chlorophyll molecules (green). Notice that the special pair and the water-oxidizing complex lay toward the thylakoid-lumen side of the membrane.
FIGURE 19.13 The structure of photosystem II. The D1 (red) and D2 (blue) subunits and the numerous bound chlorophyll molecules (green). Notice that the special pair and the water-oxidizing complex lay toward the thylakoid-lumen side of the membrane.
[Drawn from 1S5L.pdb.]
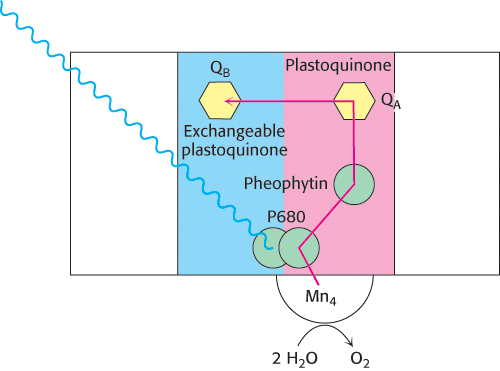
FIGURE 19.14Electron flow through photosystem II. Light absorption induces electron transfer from P680 down an electron-transfer pathway to an exchangeable plastoquinone. The positive charge on P680 is neutralized by electron flow from water molecules bound at the manganese center.
The photochemistry of photosystem II begins with excitation of a special pair of chlorophyll molecules that are bound by the D1 and D2 subunits (Figure 19.14). Because the chlorophyll a molecules of the special pair absorb light at 680 nm, the special pair is often called P680. On excitation, P680 rapidly transfers an electron to a nearby pheophytin. From there, the electron is transferred first to a tightly bound plastoquinone at site QA and then to a mobile plastoquinone at site QB. This electro flow is entirely analogous to that in the bacterial system. With the arrival of a second electron and the uptake of two protons, the mobile plastoquinone is reduced to QH2. At this point, the energy of two photons has been safely and efficiently stored in the reducing potential of QH2.
The major difference between the bacterial system and photosystem II is the source of the electrons that are used to neutralize the positive charge formed on the special pair. P680+, a very strong oxidant, extracts electrons from water molecules bound at the water-oxidizing complex (WOC), also called the manganese center. The core of this complex includes a calcium ion, four manganese ions, and four water molecules (Figure 19.15A). Manganese was apparently evolutionarily selected for this role because of its ability to exist in multiple oxidation states and to form strong bonds with oxygen-containing species. In its reduced form, the WOC oxidizes two molecules of water to form a single molecule of oxygen. Each time the absorbance of a photon powers the removal of an electron from P680, the positively charged special pair extracts an electron from a tyrosine residue (often designated Z) of subunit D1 of the WOC, forming a tyrosine radical (Figure 19.15B). The tyrosine radical then removes an electron from a manganese ion. This process occurs four times, with the result that H2O is oxidized to generate O2 and H+. Four photons must be absorbed to extract four electrons from a water molecule (Figure 19.16). The four electrons harvested from water are used to reduce two molecules of Q to QH2. All oxygenic phototrophs, the most common type of photosynthetic organism, use the same inorganic core and protein components for capturing the energy of sunlight. A single solution to the biochemical problem of extracting electrons from water evolved billions of years ago, and has been conserved for use under a wide variety of phylogenetic and ecological circumstances.

Evolution of oxygen is evident by the generation of bubbles in the aquatic plant Elodea.
[Colin Milkins/Oxford Scientific Films/Photolibrary.]
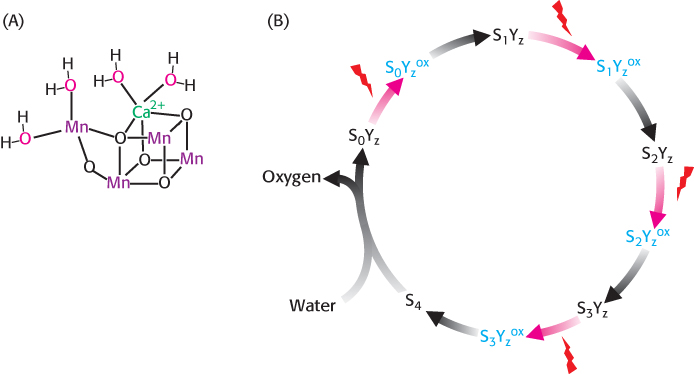
FIGURE 19.15The core of the water-oxidizing complex. (A) The deduced core structure of the water-oxidizing complex (WOC), including four manganese ions and one calcium ion, is shown. The valence states of the individual manganese ions are not indicated because of uncertainty about the charge on the individual ions. The center is oxidized, one electron at a time, until two H2O molecules are oxidized to form a molecule of O2, which is then released from the complex. (B) The absorption of photons by the reaction center generates a tyrosine radical (red arrows), which then extracts electrons from the manganese ions. The structures are designated S0 to S4 to indicate the number of electrons that have been removed.
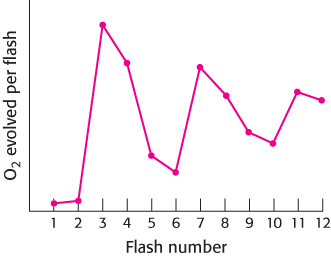
FIGURE 19.16Four photons are required to generate one oxygen molecule. When dark-adapted chloroplasts are exposed to a brief flash of light, one electron passes through photosystem II. Monitoring the O2 released after each flash reveals that four flashes are required to generate each O2 molecule. The peaks in O2 release are after the 3rd, 7th, and 11th flashes because the dark-adapted chloroplasts start in the S1 state—that is, the one-electron reduced state.
Photosystem II spans the thylakoid membrane such that the site of quinone reduction is on the side of the stroma, whereas WOC lies in the thylakoid lumen. Thus, the two protons that are taken up with the reduction of Q to QH2 come from the stroma, and the four protons that are liberated in the course of water oxidation are released into the lumen. This distribution of protons generates a proton gradient across the thylakoid membrane characterized by an excess of protons in the thylakoid lumen compared with the stroma (Figure 19.17).
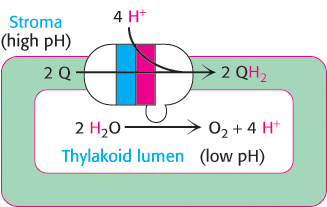
FIGURE 19.17Proton-gradient direction. Photosystem II releases protons into the thylakoid lumen and takes them up from the stroma. The result is a pH gradient across the thylakoid membrane with an excess of protons (low pH) inside.
Cytochrome bf links photosystem II to photosystem I
Electrons flow from photosystem II to photosystem I through the cytochrome bf complex. This complex catalyzes the transfer of electrons from plastoquinol (QH2) to plastocyanin (Pc), a small, soluble copper protein in the thylakoid lumen.
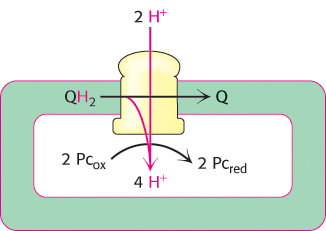
FIGURE 19.18Cytochrome bf contribution to proton gradient. The cytochrome bf complex oxidizes QH2 to Q through the Q cycle. Four protons are released into the thylakoid lumen in each cycle.
The two protons from plastoquinol are released into the thylakoid lumen. This reaction is reminiscent of that catalyzed by Complex III in oxidative phosphorylation, and most components of the cytochrome bf complex are homologous to those of Complex III. The cytochrome bf complex includes four subunits: a 23-kDa cytochrome with two b-type hemes, a 20-kDa Rieske-type Fe–S protein, a 33-kDa cytochrome f with a c-type cytochrome, and a 17-kDa chain.
This complex catalyzes the reaction by proceeding through the Q cycle (Figure 18.12). In the first half of the Q cycle, plastoquinol (QH2) is oxidized to plastoquinone (Q), one electron at a time. The electrons from plastoquinol flow through the Fe–S protein to convert oxidized plastocyanin (Pc) into its reduced form.
In the second half of the Q cycle, cytochrome bf reduces a molecule of plastoquinone from the Q pool to plastoquinol, taking up two protons from one side of the membrane, and then reoxidizes plastoquinol to release these protons on the other side. The enzyme is oriented so that protons are released into the thylakoid lumen and taken up from the stroma, contributing further to the proton gradient across the thylakoid membrane (Figure 19.18).
Photosystem I uses light energy to generate reduced ferredoxin, a powerful reductant
The final stage of the light reactions is catalyzed by photosystem I, a transmembrane complex consisting of about 15 polypeptide chains and multiple associated proteins and cofactors (Figure 19.19). The core of this system is a pair of similar subunits, psaA (83 kDa, red) and psaB (82 kDa, blue), which bind 80 chlorophyll molecules as well as other redox factors. These subunits are quite a bit larger than the core subunits of photosystem II and the bacterial reaction center. Nonetheless, they appear to be homologous; the terminal 40% of each subunit is similar to a corresponding subunit of photosystem II. A special pair of chlorophyll a molecules lie at the center of the structure and absorb light maximally at 700 nm. This center, called P700, initiates photoinduced charge separation (Figure 19.20). The electron travels from P700 down a pathway through chlorophyll at site A0 and quinone at site A1 to a set of 4Fe–4S clusters. The next step is the transfer of the electron to ferredoxin (Fd), a soluble protein containing a 2Fe–2S cluster coordinated to four cysteine residues (Figure 19.21). Ferredoxin transfers electrons to NADP+. Meanwhile, P700+ captures an electron from reduced plastocyanin provided by photosystem II to return to P700 so that P700 can be excited again. Thus, the overall reaction catalyzed by photosystem I is a simple one-electron oxidation–reduction reaction.
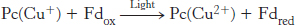
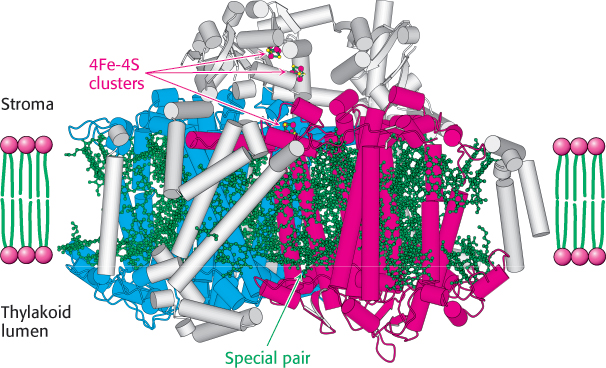
 FIGURE 19.19 The structure of photosystem I. The psaA and psaB subunits are shown in red and blue, respectively. Notice the numerous bound chlorophyll molecules, shown in green, including the special pair, as well as the iron–sulfur clusters that facilitate electron transfer from the stroma.
FIGURE 19.19 The structure of photosystem I. The psaA and psaB subunits are shown in red and blue, respectively. Notice the numerous bound chlorophyll molecules, shown in green, including the special pair, as well as the iron–sulfur clusters that facilitate electron transfer from the stroma.
[Drawn from 1JBO.pdb.]
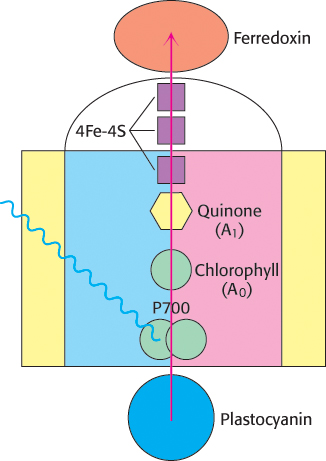
FIGURE 19.20Electron flow through photosystem I to ferredoxin. Light absorption induces electron transfer from P700 down an electron-transfer pathway that includes a chlorophyll molecule, a quinone molecule, and three 4Fe–4S clusters to reach ferredoxin. The positive charge left on P700 is neutralized by electron transfer from reduced plastocyanin.
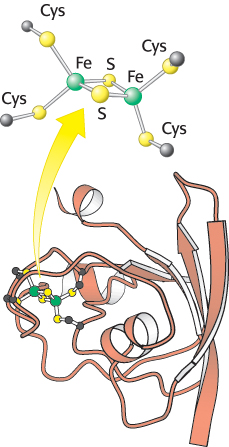
 FIGURE 19.21 Structure of ferredoxin. In plants, ferredoxin contains a 2Fe-2S cluster. This protein accepts electrons from photosystem I and carries them to ferredoxin–NADP+ reductase.
FIGURE 19.21 Structure of ferredoxin. In plants, ferredoxin contains a 2Fe-2S cluster. This protein accepts electrons from photosystem I and carries them to ferredoxin–NADP+ reductase.
[Drawn from 1FXA.pdb.]
Given that the reduction potentials for plastocyanin and ferredoxin are +0.37 V and −0.45 V, respectively, the standard free energy for this reaction is +79.1 kJ mol−1 (+18.9 kcal mol−1). This uphill reaction is driven by the absorption of a 700-nm photon, which has an energy of 171 kJ mol−1 (40.9 kcal mol−1).
Ferredoxin–NADP+ reductase converts NADP+ into NADPH
The reduced ferredoxin generated by photosystem I is a strong reductant that is used as an electron source for a variety of reactions, most notably the fixation of N2 into NH3 (Section 24.1). However, ferredoxin is not useful for driving many reactions, in part because ferredoxin carries only one available electron. In contrast, NADPH, a two-electron reductant, is a widely used electron donor in biosynthetic processes, including the reactions of the Calvin cycle (Chapter 20). How is reduced ferredoxin used to drive the reduction of NADP+ to NADPH? This reaction is catalyzed by ferredoxin–NADP+ reductase, a flavoprotein with an FAD prosthetic group (Figure 19.22A). The bound FAD moiety accepts two electrons and two protons from two molecules of reduced ferredoxin to form FADH2 (Figure 19.22B). The enzyme then transfers a hydride ion (H−) to NADP+ to form NADPH. This reaction takes place on the stromal side of the membrane. Hence, the uptake of a proton in the reduction of NADP+ further contributes to the generation of the proton gradient across the thylakoid membrane.
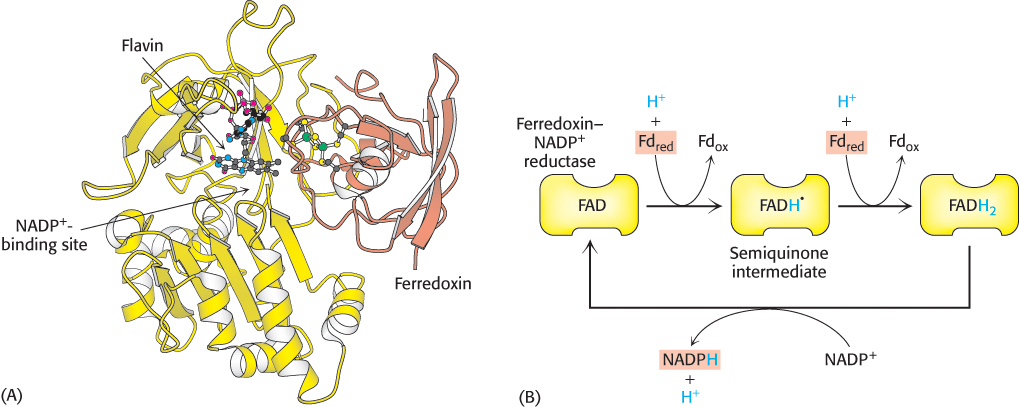
 FIGURE 19.22 Structure and function of ferredoxin–NADP+ reductase. (A) Structure of ferredoxin–NADP+ reductase. This enzyme accepts electrons, one at a time, from ferredoxin (shown in orange). (B) Ferredoxin–NADP+ reductase first accepts two electrons, generated by photosystem I, and two protons from the lumen to form two molecules of reduced ferredoxin (Fd). Reduced ferredoxin is then used to form FADH2, which then transfers two electrons and a proton to NADP+ to form NADPH in the stroma.
FIGURE 19.22 Structure and function of ferredoxin–NADP+ reductase. (A) Structure of ferredoxin–NADP+ reductase. This enzyme accepts electrons, one at a time, from ferredoxin (shown in orange). (B) Ferredoxin–NADP+ reductase first accepts two electrons, generated by photosystem I, and two protons from the lumen to form two molecules of reduced ferredoxin (Fd). Reduced ferredoxin is then used to form FADH2, which then transfers two electrons and a proton to NADP+ to form NADPH in the stroma.
[Drawn from 1EWY.pdb.]
The cooperation between photosystem I and photosystem II creates a flow of electrons from H2O to NADP+. The pathway of electron flow is called the Z scheme of photosynthesis because the redox diagram from P680 to P700* looks like the letter Z (Figure 19.23).
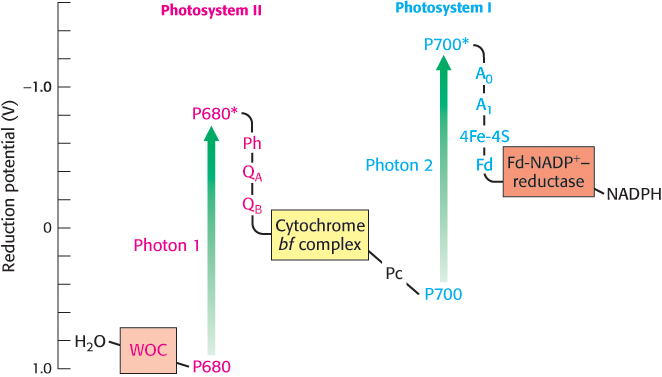
FIGURE 19.23Pathway of electron flow from H2O to NADP+ in photosynthesis. This endergonic reaction is made possible by the absorption of light by photosystem II (P680) and photosystem I (P700). Abbreviations: Ph, pheophytin; QA and QB, plastoquinone-binding proteins; Pc, plastocyanin; A0 and A1, acceptors of electrons from P700*; Fd, ferredoxin; WOC, water-oxidizing complex.

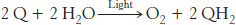
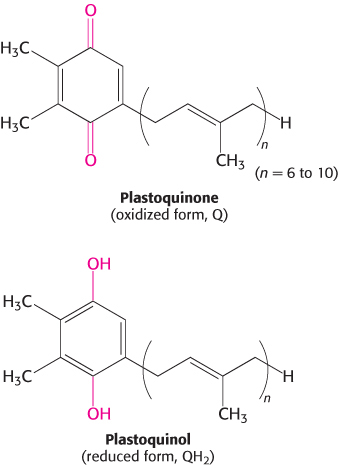

 FIGURE 19.13 The structure of photosystem II. The D1 (red) and D2 (blue) subunits and the numerous bound chlorophyll molecules (green). Notice that the special pair and the water-
FIGURE 19.13 The structure of photosystem II. The D1 (red) and D2 (blue) subunits and the numerous bound chlorophyll molecules (green). Notice that the special pair and the water-




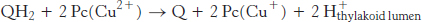

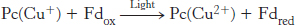

 FIGURE 19.19 The structure of photosystem I. The psaA and psaB subunits are shown in red and blue, respectively. Notice the numerous bound chlorophyll molecules, shown in green, including the special pair, as well as the iron–
FIGURE 19.19 The structure of photosystem I. The psaA and psaB subunits are shown in red and blue, respectively. Notice the numerous bound chlorophyll molecules, shown in green, including the special pair, as well as the iron–

 FIGURE 19.21 Structure of ferredoxin. In plants, ferredoxin contains a 2Fe-
FIGURE 19.21 Structure of ferredoxin. In plants, ferredoxin contains a 2Fe-
 FIGURE 19.22 Structure and function of ferredoxin–
FIGURE 19.22 Structure and function of ferredoxin–