21.3Epinephrine and Glucagon Signal the Need for Glycogen Breakdown
Epinephrine and Glucagon Signal the Need for Glycogen Breakdown
Protein kinase A activates phosphorylase kinase, which in turn activates glycogen phosphorylase. What activates protein kinase A? What is the signal that ultimately triggers an increase in glycogen breakdown?
G proteins transmit the signal for the initiation of glycogen breakdown
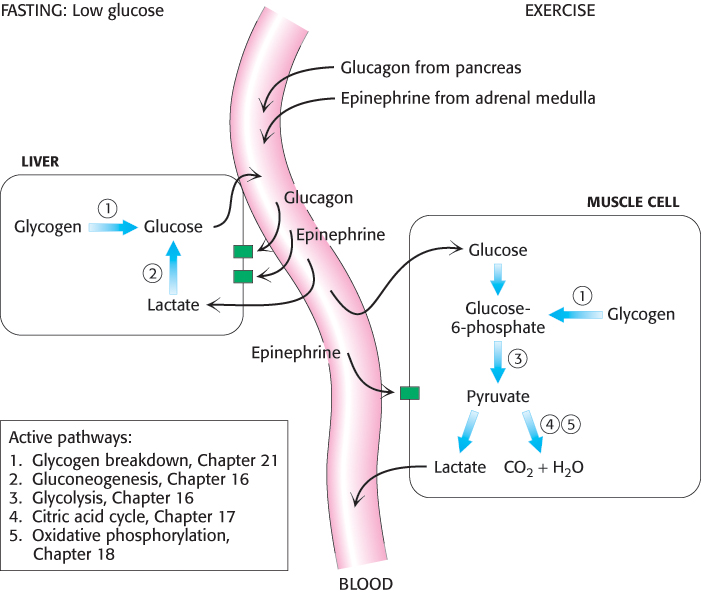
Several hormones greatly affect glycogen metabolism. Glucagon and epinephrine trigger the breakdown of glycogen. Muscular activity or its anticipation leads to the release of epinephrine (adrenaline), a catecholamine derived from tyrosine, from the adrenal medulla. Epinephrine markedly stimulates glycogen breakdown in muscle and, to a lesser extent, in the liver. The liver is more responsive to glucagon, a polypeptide hormone secreted by the α cells of the pancreas when the blood-
How do hormones trigger the breakdown of glycogen? They initiate a cyclic AMP signal-
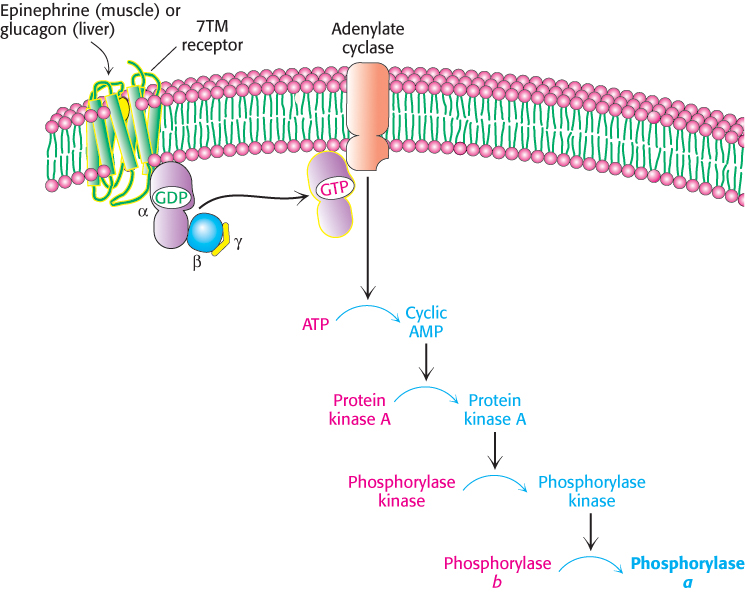
The signal molecules epinephrine and glucagon bind to specific seven-
transmembrane (7TM) receptors in the plasma membranes of target cells (Section 14.1). Epinephrine binds to the β-adrenergic receptor in muscle, whereas glucagon binds to the glucagon receptor in the liver. These binding events activate the Gs protein. A specific external signal has been transmitted into the cell through structural changes, first in the receptor and then in the G protein. 
628
The GTP-
bound subunit of Gs activates the transmembrane protein adenylate cyclase. This enzyme catalyzes the formation of the second messenger cyclic AMP (cAMP) from ATP. The elevated cytoplasmic level of cAMP activates protein kinase A (Section 10.3). The binding of cAMP to inhibitory regulatory subunits triggers their dissociation from the catalytic subunits. The free catalytic subunits are now active.
629
Protein kinase A phosphorylates phosphorylase kinase, first on the β subunit and then on the α subunit, which activates glycogen phosphorylase.
The cyclic AMP cascade highly amplifies the effects of hormones. The binding of a small number of hormone molecules to cell-
The signal-
Glycogen breakdown must be rapidly turned off when necessary
It is crucial that the high-
The signal-
The regulation of glycogen phosphorylase became more sophisticated as the enzyme evolved
 Analyses of the primary structures of glycogen phosphorylase from human beings, rats, Dictyostelium (slime mold), yeast, potatoes, and E. coli have enabled inferences to be made about the evolution of this important enzyme. The 16 residues that come into contact with glucose at the active site are identical in nearly all the enzymes. There is more variation but still substantial conservation of the 15 residues at the pyridoxal phosphate-
Analyses of the primary structures of glycogen phosphorylase from human beings, rats, Dictyostelium (slime mold), yeast, potatoes, and E. coli have enabled inferences to be made about the evolution of this important enzyme. The 16 residues that come into contact with glucose at the active site are identical in nearly all the enzymes. There is more variation but still substantial conservation of the 15 residues at the pyridoxal phosphate-
Differences arise, however, when we compare the regulatory sites. The simplest type of regulation would be feedback inhibition by glucose 6-
630