Dissociation Constants of Binding Reactions Reflect the Affinity of Interacting Molecules
The concept of equilibrium also applies to the binding of one molecule to another without covalent changes to either molecule. Many important cellular processes depend on such binding “reactions,” which involve the making and breaking of various noncovalent interactions rather than covalent bonds, as discussed above. A common example is the binding of a ligand (e.g., the hormone insulin or adrenaline) to its receptor on the surface of a cell, which triggers an intracellular signaling pathway (see Chapter 15). Another example is the binding of a protein to a specific sequence of bases in a molecule of DNA, which frequently causes the expression of a nearby gene to increase or decrease (see Chapter 9). If the equilibrium constant for a binding reaction is known, the stability of the resulting complex can be predicted.
To illustrate the general approach for determining the concentration of noncovalently associated complexes, let’s calculate the extent to which a protein (P) is bound to DNA (D), forming a protein-
P + D ⇌ PD
Most commonly, binding reactions are described in terms of the dissociation constant (Kd), which is the reciprocal of the equilibrium constant. For this binding reaction, the dissociation constant is calculated from the concentrations of the three components when they are at equilibrium by

It is worth noting that in such a binding reaction, when half of the DNA is bound to the protein ([PD] = [D]), the concentration of P is equal to Kd. The lower the Kd, the lower the concentration of P needed to bind to half of D. In other words, the lower the Kd, the tighter the binding (the higher the affinity) of P for D.
Typically, a protein’s binding to a specific DNA sequence exhibits a Kd of 10–10 M, where M symbolizes molarity, or moles per liter (mol/L). To relate the magnitude of this dissociation constant to the intracellular ratio of bound to unbound DNA, let’s consider the simple example of a bacterial cell having a volume of 1.5 × 10–15 L and containing 1 molecule of DNA and 10 molecules of the DNA-
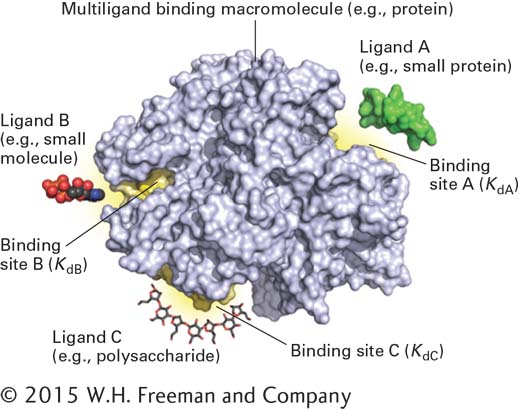
A large biological macromolecule, such as a protein, can have multiple binding surfaces for binding several molecules simultaneously (Figure 2-24). In some cases, these binding reactions are independent, with their own distinct Kd values that are independent of each other. In other cases, binding of a molecule at one site on a macromolecule can change the three-
Page 54