Fatty Acids Are Assembled from Two-Carbon Building Blocks by Several Important Enzymes
Fatty acids (see Chapter 2) play a number of important roles in cells. In addition to being a cellular fuel source (see the discussion of aerobic oxidation in Chapter 12), fatty acids are key components of both the phospholipids and the sphingolipids that make up cellular membranes; they also anchor some proteins to cellular membranes (see Figure 7-19). Thus the regulation of fatty acid synthesis plays a key role in the regulation of membrane synthesis as a whole. The major fatty acids in phospholipids contain 14, 16, 18, or 20 carbon atoms and include both saturated and unsaturated chains. The fatty acyl chains found on sphingolipids can be longer than those in the phosphoglycerides, containing up to 26 carbon atoms, and may bear other chemical modifications (e.g., hydroxylation) as well.
Fatty acids are synthesized from the two-
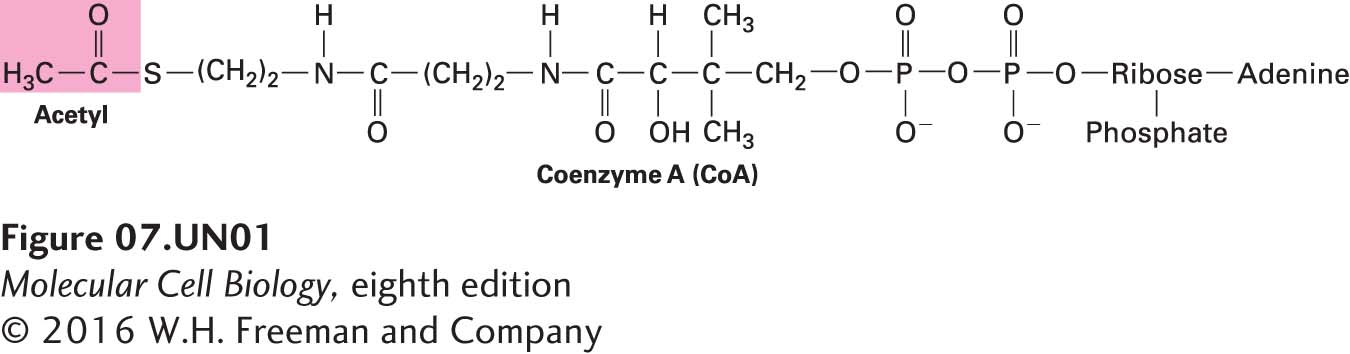
Acetyl CoA is an important intermediate in the metabolism of glucose, fatty acids, and many amino acids, as detailed in Chapter 12. It also contributes acetyl groups in many biosynthetic pathways. Saturated fatty acids (with no carbon-