CONCEPT36.5 Kidneys Adjust Water Excretion to Help Animals Maintain Homeostasis
Kidney tubules—such as the nephrons of vertebrates—typically make urine in two steps. First, fluid derived from the blood plasma enters a kidney tubule at one end. This fluid is called the primary urine. Next, the volume and composition of the fluid are modified as it flows through the remainder of the tubular system in the kidney, forming the final product, called the definitive urine, which is excreted. During this modification step, materials may be reabsorbed from the urine. Materials may also be secreted into it. Here we focus on these processes in vertebrate kidneys.
Fluid enters a nephron by ultrafiltration driven by blood pressure
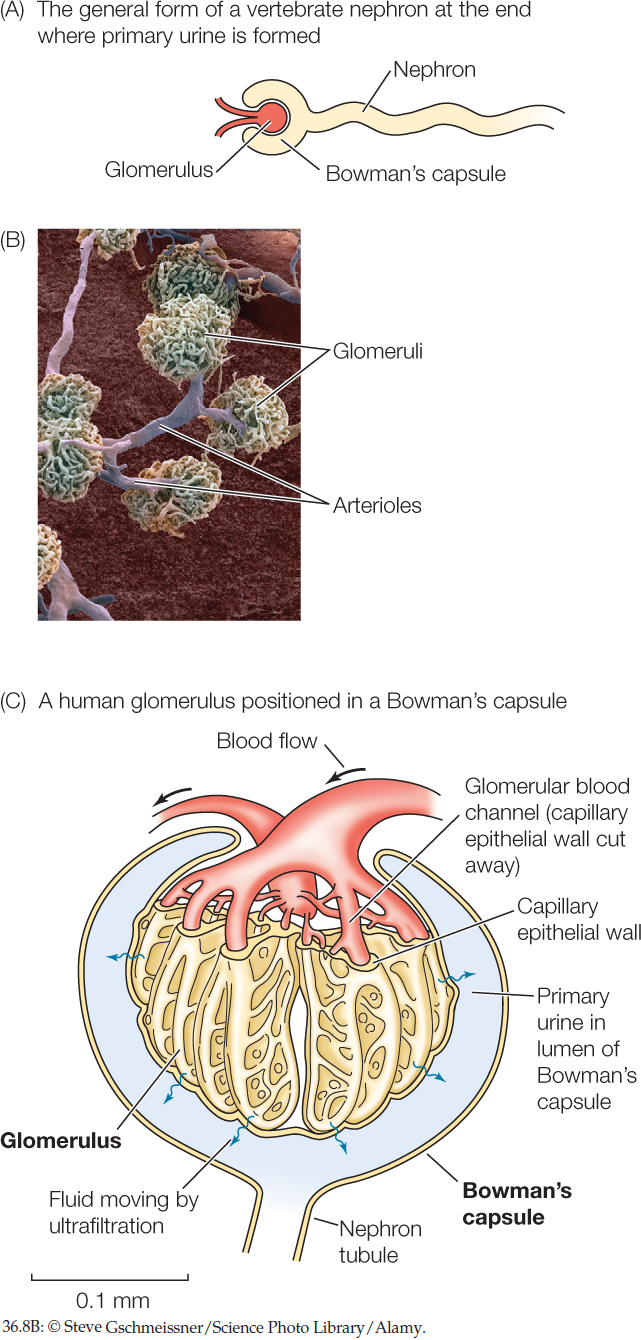
A vertebrate nephron is open at one end and closed at the other. Urine formation starts at the closed end, which consists of a cup-shaped structure called a Bowman’s capsule that encloses a dense cluster of blood capillaries called a glomerulus (plural glomeruli). The glomerulus sits within the Bowman’s capsule much like a fist pushed into an inflated balloon (FIGURE 36.8A and FIGURE 36.8B).
761
Kidneys adjust water excretion to help animals maintain homeostasis
In analyzing kidney function, an important question is whether the nephrons reabsorb substances from the tubular fluid passing through them or secrete substances into the tubular fluid. A classic way to study this question starts with measuring the glomerular filtration rate (GFR). The GFR—the volume of fluid that passes into the Bowman’s capsules of the kidneys per unit of time—can be calculated by using a marker substance (a substance artificially added to the blood) that is freely filtered out of the blood plasma (so that its concentration in the ultrafiltrate equals its plasma concentration) but not reabsorbed or secreted by the nephrons or other renal tubules. The amount of such a marker substance entering the Bowman’s capsules equals the amount leaving the body in the definitive urine. Inulin, a polysaccharide, is the most commonly used marker substance. If a different substance is reabsorbed or secreted by the nephrons or other renal tubules, the amount of that substance leaving the body in the definitive urine will be either more than or less than the amount filtered. Consider the data in the table. Assume that all three substances are freely filtered, and assume a urine flow rate of 1 milliliter (ml) per minute. As you address the questions asked, keep in mind that the amount of a substance in a solution is equal to its concentration in the solution times the volume of the solution. The concentrations of Na+ are in milliequivalents (mEq) per liter (L).
- What is the glomerular filtration rate (in ml/min)?
- Is PAH secreted or reabsorbed? Justify your answer.
- Is Na+ secreted or reabsorbed? Justify your answer.

Blood is delivered to each glomerulus at a relatively high pressure that’s sufficient to force fluid from the blood plasma to pass through minute, porelike openings in the walls of the glomerular blood capillaries and then through the interior wall of the Bowman’s capsule. This fluid enters the lumen (the open central cavity) of the Bowman’s capsule and is the primary urine (FIGURE 36.8C).
The walls of the capillaries and of the Bowman’s capsule do not allow all components of the blood to pass through. In this way, the blood is filtered as it moves through these structures. Blood cells and dissolved proteins in the plasma are filtered out and remain in the plasma. By contrast, water, salts, and small organic molecules such as glucose and amino acids pass through freely, entering the nephron. The process of forming primary urine is called ultrafiltration. The rate at which the primary urine is formed by all the nephrons functioning collectively is the glomerular filtration rate (GFR).
The primary urine is similar to the blood plasma in most ways except for lacking blood cells and proteins. The concentrations of ions (e.g., Na+ and Cl−) and of small molecules (e.g., glucose) in the primary urine are the same as those in the blood plasma. Moreover, the osmotic pressure of the primary urine is essentially equal to that of the plasma. In other words, the primary urine is isosmotic with the plasma.
As the primary urine flows through a nephron to form the definitive urine, more than half of the water and ions are typically reabsorbed into the blood plasma. More than 99 percent are reabsorbed in a person producing concentrated urine. This sounds inefficient at first. Why produce a large volume of primary urine and reabsorb most of it? The process gives the kidneys intimate access to the plasma. Humans, for example, have a GFR of about 120 milliliters per minute. At this rate the equivalent of all the plasma water in the body enters the nephrons every 30 minutes! This rate enables the kidneys to remove wastes or toxins quickly and to make rapid adjustments in the plasma volume and composition.
Many types of animals are believed to employ ultrafiltration to produce primary urine. Besides vertebrates, these animals include squid and octopuses, and crustaceans such as crayfish.
The processing of the primary urine in amphibians reveals fundamental principles of nephron function
The amphibian nephron provides a model for understanding all vertebrate nephrons. The nephrons of freshwater fish and non-avian reptiles, and most of the nephrons of birds, are similar. The parts of a vertebrate nephron are termed “early” or “proximal” if they are relatively close to the Bowman’s capsule. They are called “late” or “distal” if they are relatively far away. Along the entire length of a nephron, the nephron walls consist of a single layer of epithelial cells. However, these cells differ greatly in their structure and in the proteins they express from one part of a nephron to another.
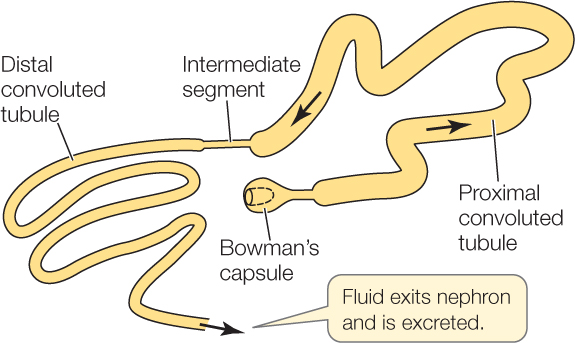
An amphibian nephron consists of two highly convoluted segments, the proximal and distal convoluted tubules:
762
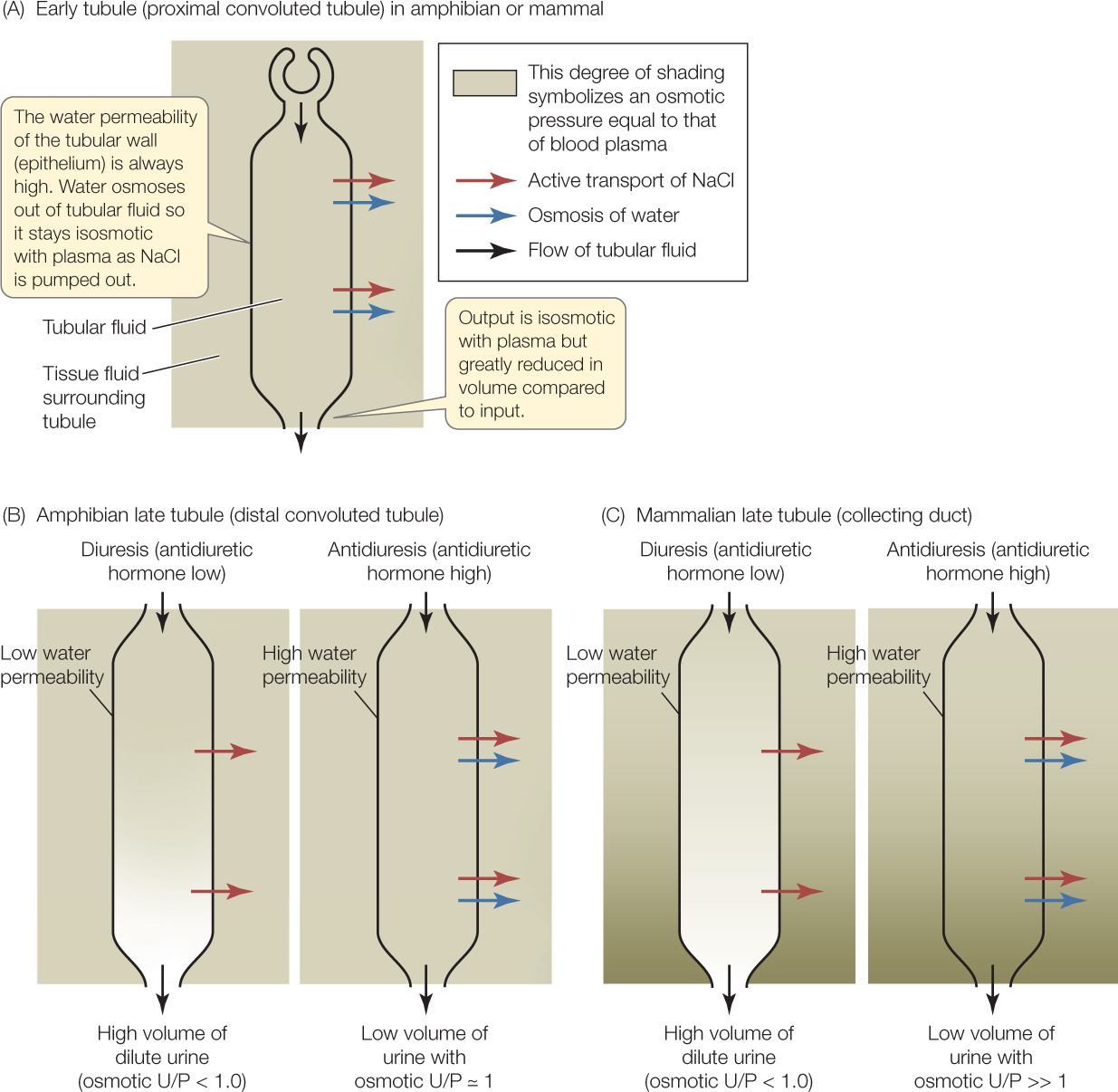
When primary urine flows out of the Bowman’s capsule, it passes first through the proximal convoluted tubule (the early part of an amphibian nephron). The nephron wall in this segment is always highly permeable to water because aquaporins (water channel proteins; see Concept 5.2) are present at all times in the cell membranes of the epithelial cells composing the wall.
As the fluid in the tubular lumen—called tubular fluid—flows through the proximal tubule, Na+ and Cl− are removed by active transport and returned to the blood. This removal of Na+ and Cl− tends to make the tubular fluid more dilute than the blood plasma. However, as the tubular fluid tends to become dilute, water also moves out of it into the nearby, more concentrated blood plasma by osmosis. The end result is that the volume of the tubular fluid is greatly reduced as the fluid flows through the proximal tubule, but the osmotic pressure of the fluid remains unchanged and equal to that of the blood plasma (FIGURE 36.9A). The epithelial cells of the proximal tubule also actively transport glucose and amino acids out of the tubular fluid, returning these valuable molecules to the blood.
Farther along the nephron, the tubular fluid enters the distal convoluted tubule. The most important difference between the proximal and distal convoluted tubules is that the wall of the distal convoluted tubule has a variable permeability to water. This is believed to be controlled by insertion and retrieval of aquaporin proteins. Aquaporin molecules in the epithelial cells of the distal tubule can either be inserted in the cell membranes or not. If they are inserted, the cells are highly permeable to water. If they are not inserted, the cells are poorly permeable to water. Like the proximal tubule, the distal tubule pumps Na+ and Cl− out of the tubular fluid by active transport.
Insertion of aquaporins is controlled by antidiuretic hormone (ADH) secreted by the hypothalamus of the brain (see Concept 35.3). Variations in ADH secretion control whether the kidney produces abundant urine—a state called diuresis—or a small amount of urine—termed antidiuresis. ADH secretion also controls the concentration of the definitive urine (FIGURE 36.9B).
When the level of ADH is high, the wall of the distal convoluted tubule (the late part of the amphibian nephron) is highly permeable to water because aquaporin molecules are inserted in the epithelial cell membranes. The distal tubule then acts much like the proximal tubule. Na+ and Cl− are pumped out of the tubular fluid, and water follows by osmosis, so the osmotic pressure in the tubular fluid remains similar to that in the blood plasma. Accordingly, the kidney is in a state of antidiuresis. It produces a low volume of urine that is approximately isosmotic with the blood plasma (see Figure 36.9B, antidiuresis).
763
By contrast, when the level of ADH is low, aquaporin molecules are not inserted in the epithelial cell membranes, and the wall of the distal tubule is poorly permeable to water. Under these conditions, water in the tubular fluid cannot readily leave it, and most of the water that enters the distal tubule passes through and is excreted. Moreover, as Na+ and Cl− are pumped out of the tubular fluid, the fluid becomes more and more dilute. The kidney is in a state of diuresis and produces a high volume of urine that is far more dilute that the blood plasma (see Figure 36.9B, diuresis).
ADH secretion is controlled in ways that help maintain homeostasis of the blood plasma. If the plasma is tending to become too dilute and full of water, sensors inform the brain, and ADH is not secreted—resulting in a high volume of dilute urine. In contrast, if the plasma is tending to become too concentrated and low in volume, ADH is secreted, and a low volume of concentrated urine is excreted.
Mammalian kidneys produce exceptionally high urine concentrations
The kidneys of amphibians regulate the urine so that the osmotic U/P ratio varies between values far less than 1.0 (urine more dilute than the blood plasma) and equal to 1.0 (urine isosmotic with the plasma). How do mammals produce exceptionally concentrated urine with a U/P ratio much higher than 1.0?
Mammalian nephrons function in ways that are fundamentally similar to those of amphibian nephrons. For example, the proximal convoluted tubule of a mammalian nephron (the early part of the nephron) is always highly permeable to water and reduces the volume of the primary urine without changing its osmotic pressure (see Figure 36.9A).
The most important difference between amphibian and mammalian kidneys is that the late tubules of the mammalian kidney—which are called collecting ducts—are surrounded with tissue fluids that in places are far more concentrated than the blood plasma. This difference explains how the mammalian kidney can produce urine that is more concentrated than the plasma. FIGURE 36.9C illustrates the mechanism. Tubular fluid travels through a collecting duct just prior to leaving the kidney as definitive urine. The tissue fluids surrounding the beginning of the collecting duct are similar to blood plasma in osmotic pressure. Along the length of the duct, however, the tissue fluids surrounding the duct have an ever-higher osmotic pressure, reaching an osmotic pressure far higher than that of the blood plasma, as Figure 36.9C shows. Na+, Cl−, and other salts cannot diffuse readily across the wall of a collecting duct, although they can cross by means of ion pumping when pumps are active.
When the ADH level is high, the wall of the collecting duct is highly permeable to water because aquaporins are inserted in the cell membranes of the epithelial cells composing the wall. As tubular fluid flows through the collecting duct, it loses water by osmosis into the concentrated tissue fluids surrounding the duct (and the blood then picks up this water). This process of water removal concentrates the salts in the tubular fluid (see Figure 36.9C, antidiuresis). Just before the tubular fluid leaves the collecting duct to enter the ureter and travel to the bladder, the tubular fluid comes to osmotic equilibrium with the tissue fluids of highest concentration. The osmotic pressure of the urine is thus far higher than that of the blood plasma. A low volume of highly concentrated urine is produced.
When the ADH level is low, the wall of the collecting duct has a low permeability to water because aquaporins are removed from the cell membranes of the epithelial cells composing the wall (see Figure 36.9C, diuresis). As tubular fluid flows through the collecting duct, it does not lose much water by osmosis into the surrounding tissue fluid. Moreover, Na+ and Cl− are actively transported out of the tubular fluid, making the fluid more and more dilute as it moves through the collecting duct. A high volume of dilute urine is produced.
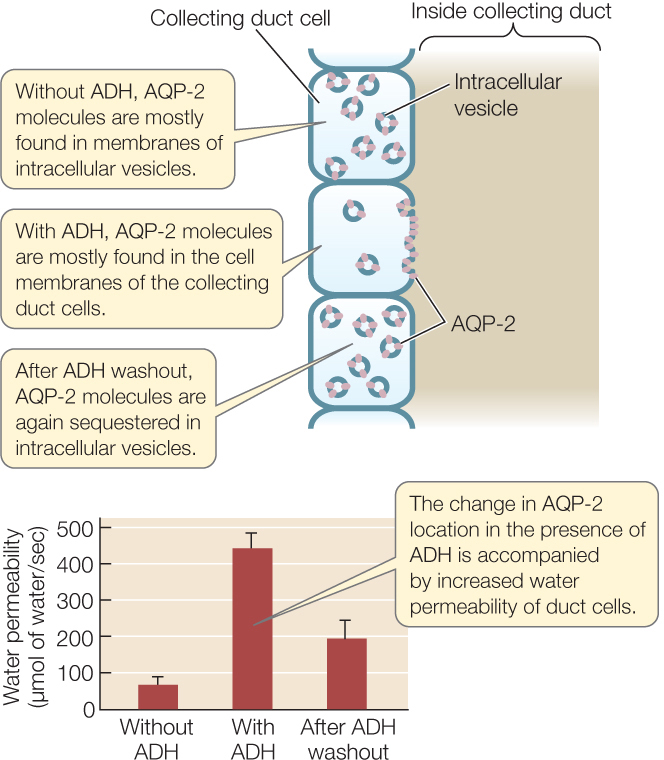
AQP-2 is the specific molecular form of aquaporin that is of greatest importance in controlling the water permeability of the collecting duct. Experiments have documented its important role (FIGURE 36.10).
Investigation
HYPOTHESIS
ADH controls water permeability by changing the cellular location of aquaporin molecules.
- Isolate collecting ducts from rat kidney.
- Use immunochemical staining to localize AQP-2 molecules in collecting duct cells without ADH, then with ADH, and—finally—after the applied ADH is washed away.
- Measure the water permeability of the collecting duct cells under the same three conditions.
CONCLUSION
In the absence of ADH, AQP-2 water channel molecules are sequestered intracellularly. When ADH is present, these water channels are inserted into the cell membranes, making the cells more permeable to water.
ANALYZE THE DATA
The results showed that ADH controls the location of AQP-2 molecules in collecting duct cells. Does ADH also affect the amount of AQP-2? The Brattleboro strain of rats does not produce ADH, and the DI strain of mice has a defect in the signaling pathway of the ADH receptor. The amount of AQP-2 produced in response to ADH and to an ADH receptor antagonist was studied in these animals. Values were normalized to baseline amounts in normal animals.
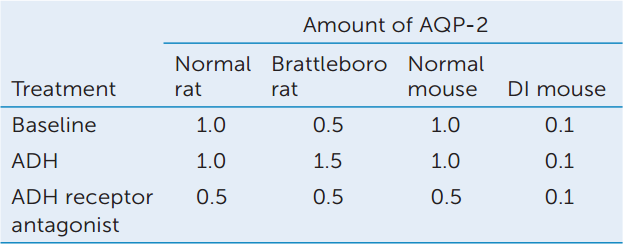
- How does ADH affect the amount of AQP-2 in normal and Brattleboro rats? Explain the difference in the responses of the two rat strains.
- Why does ADH not have an effect on the amount of AQP-2 in DI mice?
- Considering these data as a whole, construct a hypothesis to explain why Brattleboro rats can maintain AQP-2 levels at 50% of normal under baseline conditions. How would you test your hypothesis?
aK. Fushimi et al. 1993. Nature 361: 549–552; S. Nielsen et al. 1995. Proceedings of the National Academy of Sciences 92: 1013–1017.
How are the tissue fluids bathing the collecting ducts deep inside the kidney made highly concentrated? In a mammalian kidney, a portion of each nephron functions to increase the osmotic pressure of the tissue fluids surrounding it. This portion is a long, hairpin-shaped segment called the loop of Henle (pronounced Hen-lee), positioned immediately after the proximal convoluted tubule (FIGURE 36.11). All the nephrons in a mammalian kidney are organized so that their loops of Henle are parallel. This microscopic structure gives the entire kidney a visible structure. Each kidney has an outer layer, the renal cortex, where the Bowman’s capsules and the convoluted tubules of the nephrons are found. Inside this cortical layer, the kidney has an inner layer called the renal medulla. This inner layer is composed of the loops of Henle and collecting ducts (see Figure 36.11). Parts of the medulla are said to be “deeper,” the farther they are from the cortex.
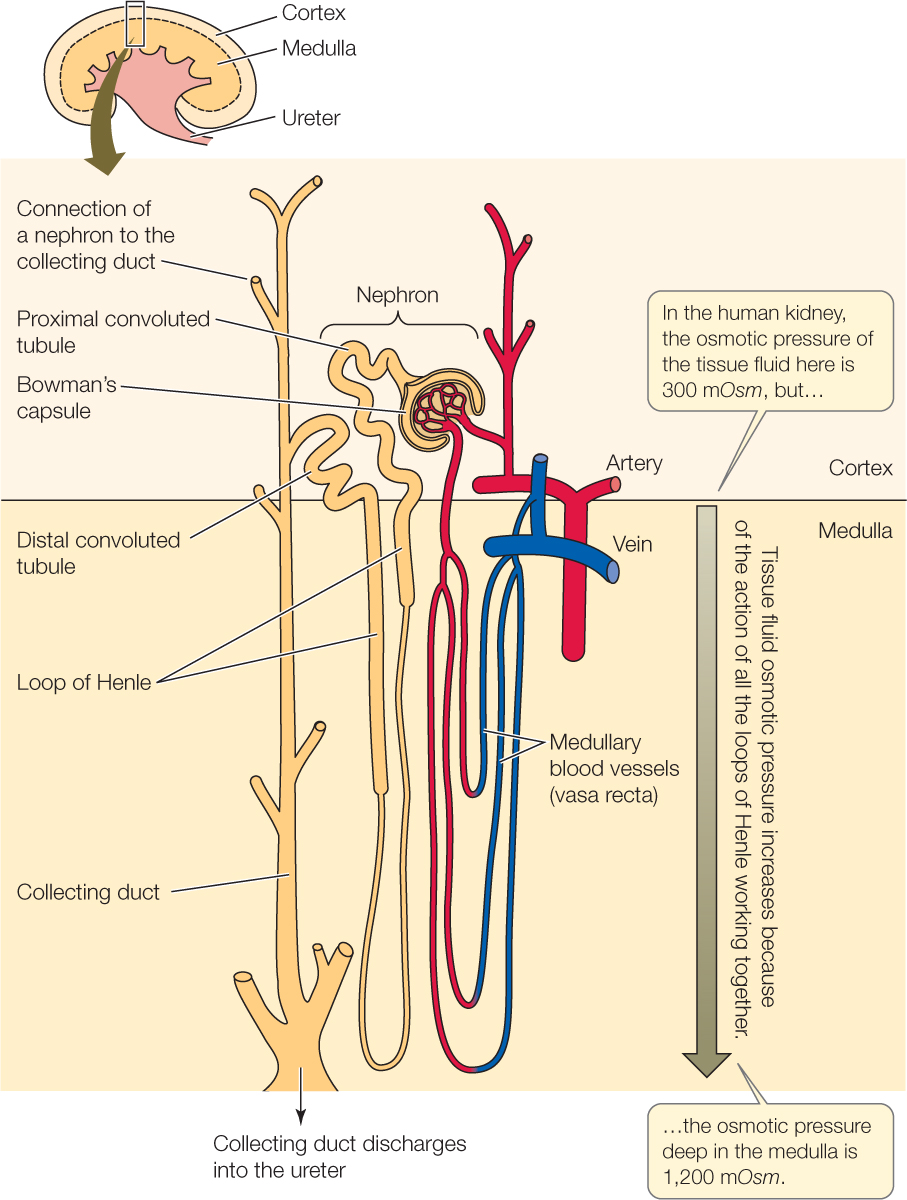
The loops of Henle in a kidney work together to increase the osmotic pressure of the tissue fluids deep in the medulla. In this way, the loops create a gradient in the osmotic pressure of the medullary tissue fluids, so that the tissue fluids near the cortex are isosmotic with the blood plasma, but those deep in the medulla are far more concentrated. For example, in the human kidney, the osmotic pressure of the tissue fluids increases from about 300 mOsm near the cortex to about 1,200 mOsm—four times the blood osmotic pressure—in the deepest medullary tissue. To create this gradient, the loops of Henle use a mechanism called countercurrent multiplication (not to be confused with countercurrent exchange) in which the flow of tubular fluid in opposite directions in each loop (first into the medulla and then out) multiplies effects of active ion transport. The loops of Henle do not themselves concentrate the urine, but they set the stage for the urine to be concentrated in the collecting ducts.
764
Each nephron empties into a collecting duct, and the fluid from the nephron then flows through the collecting duct from the cortex to deep in the medulla (see Figure 36.11). As the fluid flows through the collecting duct, it becomes definitive urine, which exits the collecting duct to flow to the bladder via the ureter draining the kidney (see Figure 36.2). Figure 36.9C, which we have already discussed, shows how the volume and concentration of the definitive urine are determined in the collecting duct, both when the ADH level is high and when it is low.
Birds differ from mammals in that only 10–30 percent of their nephrons have loops of Henle. This limits the concentrating ability of bird kidneys relative to mammalian kidneys.
The Malpighian tubules of insects employ a secretory mechanism of producing primary urine
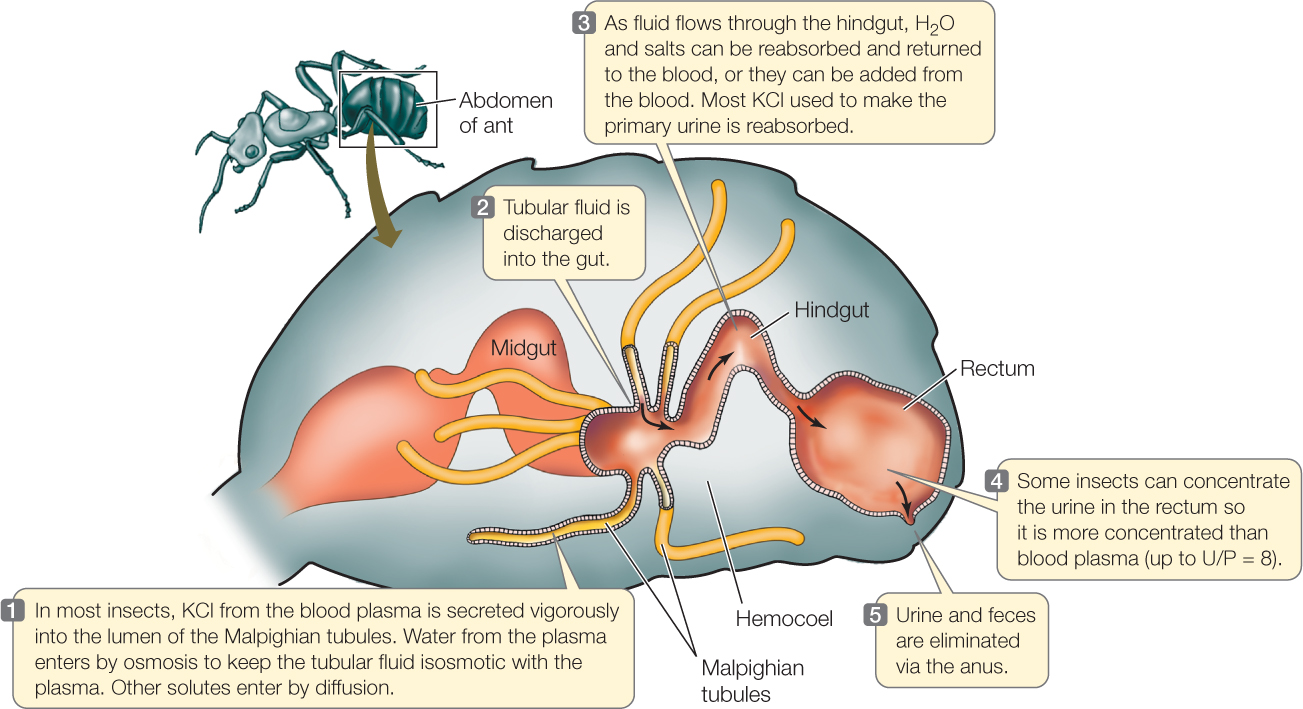
The primary urine of insects is produced in blind-ended tubules, the Malpighian tubules, that open into the gut between the midgut and hindgut (FIGURE 36.12). Insects have a low-pressure, open circulatory system (see Concept 32.1) and do not use high blood pressure to cause ultrafiltration into the tubules. Instead, the epithelial cells in the tubule walls employ active transport to secrete KCl (or in some cases other salts) at a high rate into the tubular fluid, giving the tubular fluid a high osmotic pressure. The walls of the tubules are permeable to water. Water therefore enters the tubules by osmosis from the more dilute blood surrounding them, creating the primary urine. Other salts besides KCl enter by diffusion. The process is described as a “secretory mechanism” for producing primary urine.
765
766
The primary urine flows out of the Malpighian tubules into the hindgut and then flows through the hindgut, which greatly modifies the volume and composition of the fluid before it is excreted. Thus the excretory system consists of both the Malpighian tubules and the hindgut. One important function of the hindgut is to reabsorb the KCl used to produce primary urine, so the KCl can be used again in urine production.
CHECKpointCONCEPT36.5
- Human patients suffering from heart failure often become unable to produce adequate amounts of urine, a condition known as kidney failure. How could heart failure lead to kidney failure?
- What is the difference between an ultrafiltration-based mechanism of producing primary urine and a secretion-based mechanism?
- Antidiuretic hormone (ADH) has the same effect on the late tubules of both amphibians and mammals: it increases the water permeability of epithelial cells that are otherwise poorly permeable to water. If the effect is the same, why do amphibian kidneys produce urine of the same osmotic pressure as the blood plasma when the ADH concentration is high, whereas mammals produce urine more concentrated than blood plasma?
How can a salmon keep the composition of its blood plasma stable regardless of whether it is swimming in seawater or fresh water?
ANSWER When a salmon travels from seawater into fresh water, it reverses many of the processes it employs to maintain water and salt homeostasis. In seawater, cells in the gill epithelium actively transport Na+ and Cl− out of the blood plasma into the surrounding seawater (Concept 36.3). When a salmon enters fresh water, its gills reverse their direction of active ion transport, pumping Na+ and Cl− into the blood plasma from the surrounding river or pond water.
Drinking behavior also reverses. When in seawater, a salmon drinks to replace the water it steadily loses by osmosis across its external body surfaces. When the fish enters fresh water, it stops drinking.
Kidney function reverses as well. When a salmon is in seawater, its kidneys produce just a small amount of urine per day, and the urine has the same osmotic pressure as the blood plasma. This concentration, although maximal for a fish, is inadequate to keep the plasma concentration from rising. As a result, the gills must bear most of the responsibility for voiding excess ions in a way that will maintain the plasma hyposmotic to seawater. When a salmon enters fresh water, its kidneys switch to producing a large volume of dilute urine.