34.3
Diversity Is Generated by Gene Rearrangements
A mammal such as a mouse or a human being can synthesize large amounts of specific antibody against virtually any foreign determinant within a matter of days after exposure. We have seen that antibody specificity is determined by the amino acid sequences of the variable regions of both light and heavy chains, which brings us to the key question: How are different variable-region sequences generated?
The discovery of distinct variable and constant regions in the L and H chains raised the possibility that the genes that encode immunoglobulins have an unusual architecture that facilitates the generation of a diverse set of polypeptide products. In 1965, William Dreyer and Claude Bennett proposed that multiple V (variable) genes are separate from a single C (constant) gene in embryonic (germ-line) DNA. According to their model, one of these V genes becomes joined to the C gene in the course of differentiation of the antibody-producing cell. A critical test of this novel hypothesis had to await the isolation of pure immunoglobulin mRNA and the development of techniques for analyzing mammalian genomes. Twenty years later, Susumu Tonegawa found that V and C genes are indeed far apart in embryonic DNA but are closely associated in the DNA of antibody-producing cells. Thus, immunoglobulin genes are rearranged in the differentiation of lymphocytes.
J (joining) genes and D (diversity) genes increase antibody diversity

FIGURE 34.17The κ light-chain locus. This part of human chromosome 2 includes an array of 40 segments that encode the variable (V) region (approximately residues 1–97) of the light chain, an array of 5 segments that encode the joining (J) region (residues 98–110), and a single region that encodes the constant (C) region.
Sequencing studies carried out by Susumu Tonegawa, Philip Leder, and Leroy Hood revealed that V genes in embryonic cells do not encode the entire variable region of L and H chains. Consider, for example, the region that encodes the κ light-chain family. A tandem array of 40 segments, each of which encodes approximately the first 97 residues of the variable domain of the L chain, is present on human chromosome 2 (Figure 34.17).
However, the variable region of the L chain contains 110 residues. Where is the DNA that encodes the last 13 residues of the variable domain? For L chains in undifferentiated cells, this stretch of DNA is located in an unexpected place: near the C gene. It is called the J gene because it joins the V and C gene segments in a differentiated cell. In fact, a tandem array of five J gene segments is located near the C gene in embryonic cells. In the differentiation of an antibody-producing cell, a V gene segment is joined to a J gene segment to form a complete gene for the variable region (Figure 34.18). RNA splicing (Section 29.3) generates an mRNA molecule for the complete L chain by linking the coding regions for the rearranged VJ unit with that for the C unit (Figure 34.19).
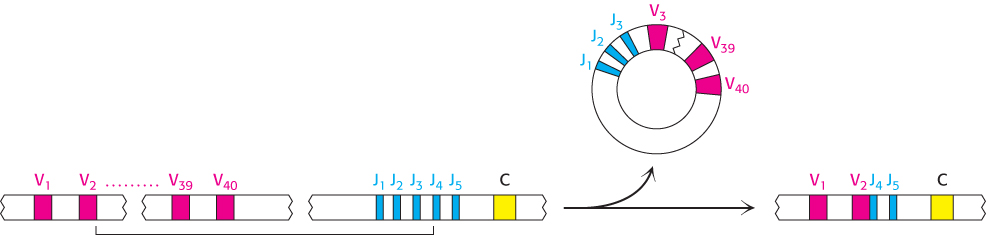
FIGURE 34.18VJ recombination. A single V gene (in this case, V2) is linked to a J gene (here, J4) to form an intact VJ region. The intervening DNA is released in a circular form. Because the V and J regions are selected at random and the joint between them is not always in exactly the same place, many VJ combinations can be generated by this process.
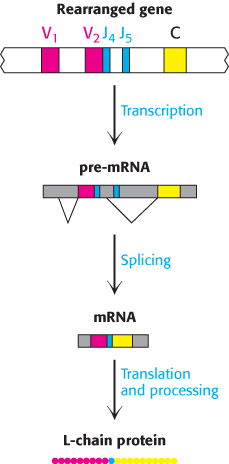
FIGURE 34.19Light-chain expression. The light-chain protein is expressed by the transcription of the rearranged gene to produce a pre-RNA molecule with the VJ and C regions separated. RNA splicing removes the intervening sequences to produce an mRNA molecule with the VJ and C regions linked. Translation of the mRNA and processing of the initial protein product produce the light chain.
J gene segments are important contributors to antibody diversity because they encode part of the last hypervariable segment (CDR3). In forming a continuous variable-region gene, any of the 40 V gene segments can become linked to any of 5 J gene segments. Thus, somatic recombination of these gene segments amplifies the diversity already present in the germ line. The linkage between V and J is not precisely controlled. Recombination between these genes can take place at one of several bases near the codon for residue 95, generating additional diversity. A similar array of V and J gene segments encoding the λ light chain is present on human chromosome 22. This region includes 30 Vλ gene segments and four Jλ gene segments. In addition, this region includes four distinct C genes, in contrast with the single C gene in the κ locus.
In humans, the genes encoding the heavy chain are present on chromosome 14. Remarkably, the variable domain of heavy chains is assembled from three rather than two segments. In addition to VH gene segments that encode residues 1 through 94 and JH gene segments that encode residues 98 through 113, this chromosomal region includes a distinct set of segments that encode residues 95 through 97 (Figure 34.20). These gene segments are called D (for diversity). Some 27 D gene segments lie between 51 VH and 6 JH gene segments. The recombination process first joins a D gene segment to a JH gene segment; a VH gene segment is then joined to DJH. A greater variety of antigen-binding patches and clefts can be formed by the H chain than by the L chain because the H chain is encoded by three rather than two gene segments. Moreover, CDR3 of the H chain is diversified by the action of terminal deoxyribonucleotidyl transferase, a special DNA polymerase that requires no template. This enzyme inserts extra nucleotides between VH and D. The V(D)J recombination of both the L and the H chains is executed by specific enzymes present in immune cells. These proteins, called RAG-1 and RAG-2, recognize specific DNA sequences called recombination signal sequences (RSSs) adjacent to the V, D, and J gene segments and facilitate the cleavage and religation of the DNA segments.
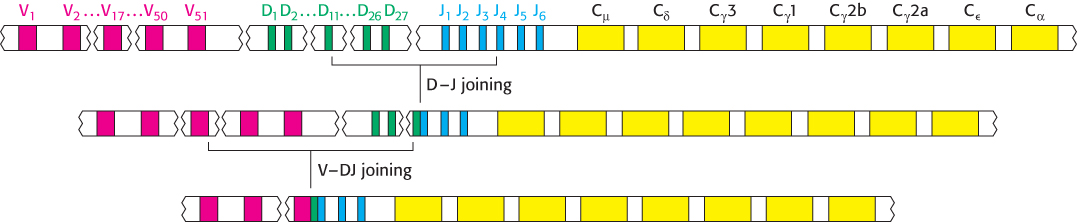
FIGURE 34.20V(D)J recombination. The heavy-chain locus includes an array of 51 V segments, 27 D segments, and 6 J segments. Gene rearrangement begins with D–J joining, followed by further rearrangement to link the V segment to the DJ segment.
More than 108 antibodies can be formed by combinatorial association and somatic mutation
Let us recapitulate the sources of antibody diversity. The germ line contains a rather large repertoire of variable-region genes. For κ light chains, there are about 40 V-segment genes and 5 J-segment genes. Hence, a total of 40 × 5 = 200 kinds of complete Vκ genes can be formed by the combinations of V and J. A similar analysis suggests that at least 120 different λ light chains can be generated. A larger number of heavy-chain genes can be formed because of the role of the D segments. For 51 V, 27 D, and 6 J gene segments, the number of complete VH genes that can be formed is 8262. The association of 320 kinds of L chains with 8262 kinds of H chains would yield 2.6 × 106 different antibodies. Variability in the exact points of segment joining and other mechanisms increases this value by at least two orders of magnitude.
Even more diversity is introduced into antibody chains by somatic mutation—that is, by the introduction of mutations into the recombined genes. In fact, a 1000-fold increase in binding affinity is seen in the course of a typical humoral immune response, arising from somatic mutation, a process called affinity maturation. The generation of an expanded repertoire leads to the selection of antibodies that more precisely fit the antigen. Thus, nature draws on each of three sources of diversity—a germ-line repertoire, somatic recombination, and somatic mutation—to form the rich variety of antibodies that protect an organism from foreign incursions.
The oligomerization of antibodies expressed on the surfaces of immature B cells triggers antibody secretion
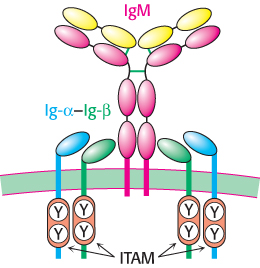
FIGURE 34.21B-cell receptor. This complex consists of a membrane-bound IgM molecule noncovalently bound to two Ig-α–Ig-β heterodimers. The intracellular domains of each of the Ig-α and Ig-β chains include an immunoreceptor tyrosine-based activation motif (ITAM).
The processes heretofore described generate a highly diverse set of antibody molecules—a key first step in the generation of an immune response. The next stage is the selection of a particular set of antibodies directed against a specific invader. How is this selection accomplished? Each immature B cell, produced in the bone marrow, expresses a specific monomeric form of IgM attached to its surface (Figure 34.21). Each cell expresses approximately 105 IgM molecules, but all of these molecules are identical in amino acid sequence and, hence, in antigen-binding specificity. Thus, the selection of a particular immature B cell for growth will lead to the amplification of an antibody with a unique specificity. The selection process begins with the binding of an antigen to the membrane-bound antibody.
Associated with each membrane-linked IgM molecule are two molecules of a heterodimeric membrane protein called Ig-α–Ig-β (Figure 34.21). Examination of the amino acid sequences of Ig-α and Ig-β is highly instructive. The amino terminus of each protein lies outside the cell and corresponds to a single immunoglobulin fold, and the carboxyl terminus, which lies inside the cell, includes a sequence of 18 amino acids called an immunoreceptor tyrosine-based activation motif (ITAM; Figure 34.21). As its name suggests, each ITAM includes key tyrosine residues, which are subject to phosphorylation by particular protein kinases present in immune-system cells.
A fundamental observation linking the binding of antigen to membrane-bound antibody with the subsequent initiation of an immune response is that oligomerization, or clustering, of the antibody molecules is required (Figure 34.22). The requirement for oligomerization is reminiscent of the dimerization of receptors triggered by epidermal growth factor and insulin encountered in Section 14.3; indeed, the associated signaling mechanisms appear to be quite similar. The oligomerization of the membrane-bound antibodies results in the phosphorylation of the tyrosine residues within the ITAMs by protein tyrosine kinases including Lyn, a homolog of Src (Section 14.5). The phosphorylated ITAMs serve as docking sites for a protein kinase termed spleen tyrosine kinase (Syk), which has two SH2 domains that interact with the pair of phosphorylated tyrosine residues in each ITAM. Docked Syk adopts an active confirmation, which then phosphorylates other signal-transduction proteins, including an inhibitory subunit of the transcription factor NF-κB and an isoform of phospholipase C. The signaling processes continue downstream to activate gene expression, leading to the stimulation of cell growth and initiating further B-cell differentiation.
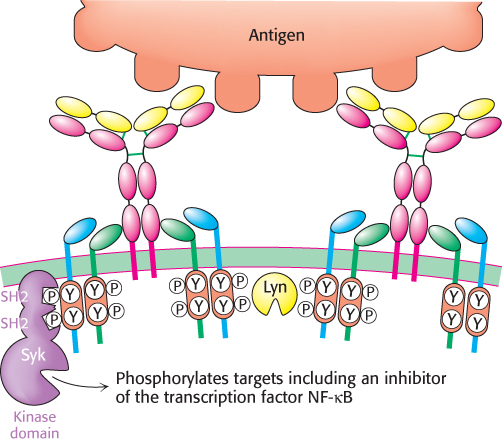
FIGURE 34.22B-cell activation. The binding of a multivalent antigen such as a bacterial or viral surface links membrane-bound IgM molecules. This oligomerization triggers the phosphorylation of tyrosine residues in the ITAM sequences by protein tyrosine kinases such as Lyn. After phosphorylation, the ITAMs serve as docking sites for Syk, a protein kinase that phosphorylates a number of targets, including transcription factors.
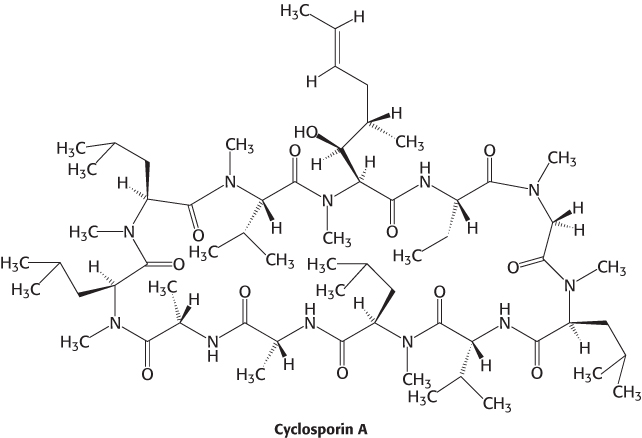
 Drugs that modulate the immune system have served as sources of insight into immune-system signaling pathways. For example, cyclosporine, a powerful suppressor of the immune system, inhibits calcineurin, a phosphatase which normally activates a transcription factor called NF-AT by dephosphorylating it.
Drugs that modulate the immune system have served as sources of insight into immune-system signaling pathways. For example, cyclosporine, a powerful suppressor of the immune system, inhibits calcineurin, a phosphatase which normally activates a transcription factor called NF-AT by dephosphorylating it.
The potent inhibition of the immune system, or immunosuppression, induced by cyclosporine reveals how crucial the activity of NF-AT is to the development of an immune response. Without such drugs, organ transplantation would be extremely difficult because transplanted tissue expresses a wide range of foreign antigens, causing the host immune system to reject the new tissue.
The role of oligomerization in the B-cell signaling pathway is illuminated when we consider the nature of many antigens presented by pathogens. The surfaces of many viruses, bacteria, and parasites are characterized by arrays of identical membrane proteins or membrane-linked carbohydrates. Thus, most pathogens present multiple binding surfaces that will naturally cause membrane-associated antibodies to oligomerize as they bind adjacent epitopes. In addition, the mechanism accounts for the observation that most small molecules do not induce an immune response; however, coupling multiple copies of a small molecule to a large oligomeric protein such as keyhole limpet hemocyanin (KLH), which has a molecular mass of 1 million daltons or more, promotes antibody oligomerization and, hence, the production of antibodies against the small-molecule epitope. The large protein is called the carrier of the attached chemical group, which is called a haptenic determinant. The small foreign molecule by itself is called a hapten. Antibodies elicited by attached haptens will bind unattached haptens as well.
Different classes of antibodies are formed by the hopping of VH genes
The development of an effective antibody-based immune response depends on the secretion into the blood of antibodies that have appropriate effector functions. At the beginning of this response, an alternative mRNA-splicing pathway is activated so that the production of membrane-linked IgM is supplanted by the synthesis of secreted IgM. As noted in Section 34.1, secreted IgM is pentameric and has a high avidity for antigens containing multiple identical epitopes. Later, the antibody-producing cell makes either IgG, IgA, IgD, or IgE of the same specificity as that of the initially secreted IgM. In this switch, the light chain and the variable region of the heavy chain are unchanged. Only the constant region of the heavy chain changes. This step in the differentiation of an antibody-producing cell is called class switching (Figure 34.23). In undifferentiated cells, the genes for the constant region of each class of heavy chain, called Cμ, Cδ, Cγ, Cε, and Cα, are next to one another. There are eight in all, including four genes for the constant regions of γ chains. A complete transcript for the heavy chain of an IgM antibody is formed by the translocation of a VH gene segment to a DJH gene segment within the genomic DNA (Figure 34.20), followed by RNA splicing of the VHDJH segment to the Cμ segment, analogous to what is shown in Figure 34.19.
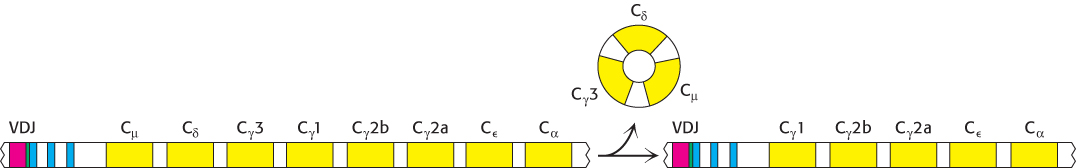
FIGURE 34.23Class switching. Further rearrangement of the heavy-chain locus results in the generation of genes for antibody classes other than IgM. In the case shown, rearrangement places the VDJ region next to the Cγ1 region, resulting in the production of IgG1. Note that no further rearrangement of the VDJ region takes place, and so the specificity of the antibody is not affected.
How are other heavy chains formed? Class switching is mediated by a gene-rearrangement process that moves a VDJ gene from a site near one C gene to a site near another C gene. Importantly, the antigen-binding specificity is conserved in class switching because the entire VHDJH gene is translocated in an intact form. For example, the antigen-combining specificity of IgA produced by a particular cell is the same as that of IgM synthesized at an earlier stage of its development. The biological significance of CH switching is that a whole recognition domain (the variable domain) is shifted from the early constant region (Cμ) to one of several other constant regions that mediate different effector functions.






 Drugs that modulate the immune system have served as sources of insight into immune-
Drugs that modulate the immune system have served as sources of insight into immune-