Antibodies to specific proteins can be generated
Immunological techniques begin with the generation of antibodies to a particular protein. An antibody (also called an immunoglobulin, Ig) is itself a protein (Figure 3.17); it is synthesized by vertebrates in response to the presence of a foreign substance, called an antigen. Antibodies have specific and high affinity for the antigens that elicited their synthesis. The binding of antibody to antigen is a step in the immune response that protects the animal from infection (Chapter 34). Foreign proteins, polysaccharides, and nucleic acids can be antigens. Small foreign molecules, such as synthetic peptides, also can elicit antibodies, provided that the small molecule is attached to a macromolecular carrier. An antibody recognizes a specific group or cluster of amino acids on the target molecule called an antigenic determinant or epitope. The specificity of the antibody–antigen interaction is a consequence of the shape complementarity between the two surfaces (Figure 3.18). Animals have a very large repertoire of antibody-producing cells, each producing an antibody that contains a unique surface for antigen recognition. When an antigen is introduced into an animal, it is recognized by a select few cells from this population, stimulating the proliferation of these cells. This process ensures that more antibodies of the appropriate specificity are produced.
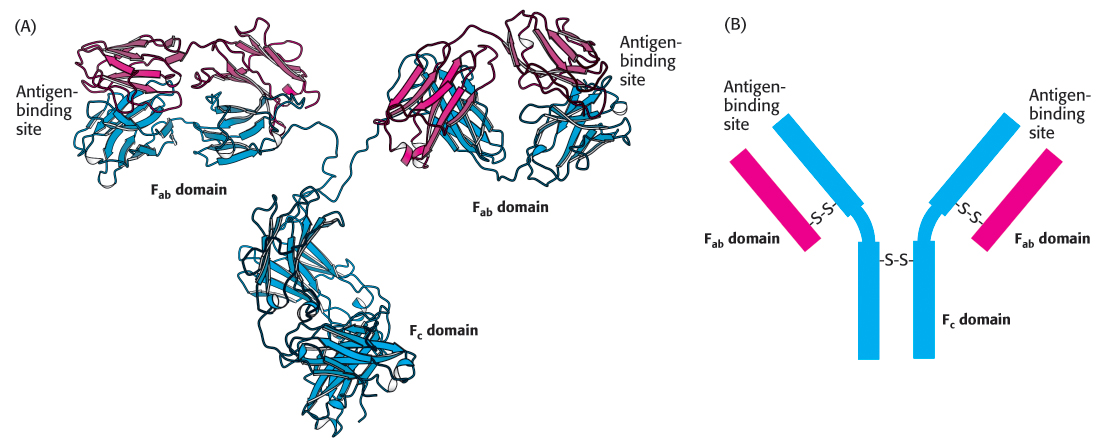
FIGURE 3.17Antibody structure. (A) Immunoglobulin G (IgG) consists of four chains, two heavy chains (blue) and two light chains (red), linked by disulfide bonds. The heavy and light chains come together to form Fab domains, which have the antigen-binding sites at the ends. The two heavy chains form the Fc domain. Notice that the Fab domains are linked to the Fc domain by flexible linkers. (B) A more schematic representation of an IgG molecule.
[Drawn from 1IGT.pdb.]
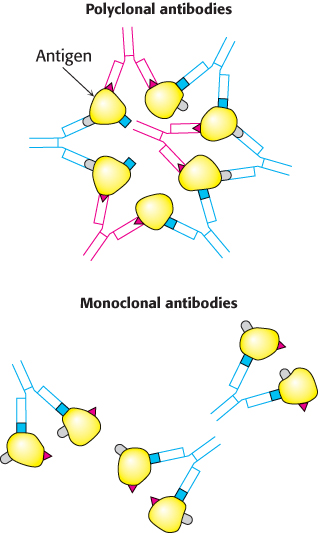
FIGURE 3.19Polyclonal and monoclonal antibodies. Most antigens have several epitopes. Polyclonal antibodies are heterogeneous mixtures of antibodies, each specific for one of the various epitopes on an antigen. Monoclonal antibodies are all identical, produced by clones of a single antibody-producing cell. They recognize one specific epitope.
[Information from R. A. Goldsby, T. J. Kindt, and B. A. Osborne, Kuby Immunology, 4th ed. (W. H. Freeman and Company, 2000), p. 154.]
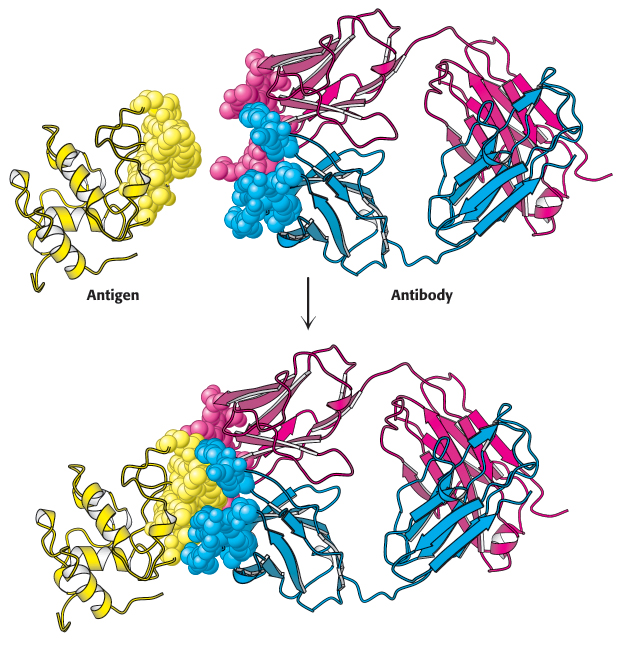
FIGURE 3.18Antigen–antibody interactions. A protein antigen, in this case lysozyme, binds to the end of an Fab domain of an antibody. Notice that the end of the antibody and the antigen have complementary shapes, allowing a large amount of surface to be buried on binding.
[Drawn from 1YQV.pdb.]
Immunological techniques depend on the ability to generate antibodies to a specific antigen. To obtain antibodies that recognize a particular protein, a biochemist injects the protein into a rabbit twice, 3 weeks apart. The injected protein acts as an antigen, stimulating the reproduction of cells producing antibodies that recognize it. Blood is drawn from the immunized rabbit several weeks later and centrifuged to separate blood cells from the supernatant, or serum. The serum, called an antiserum, contains antibodies to all antigens to which the rabbit has been exposed. Only some of them will be antibodies to the injected protein. Moreover, antibodies that recognize a particular antigen are not a single molecular species. For instance, 2,4-dinitrophenol (DNP) was used as an antigen to generate antibodies. Analyses of anti-DNP antibodies revealed a wide range of binding affinities; the dissociation constants ranged from about 0.1 nM to 1 μM. Correspondingly, a large number of bands were evident when anti-DNP antibody was subjected to isoelectric focusing. These results indicate that cells are producing many different antibodies, each recognizing a different surface feature of the same antigen. These antibodies are termed polyclonal, referring to the fact that they are derived from multiple antibody-producing cell populations (Figure 3.19). The heterogeneity of polyclonal antibodies can be advantageous for certain applications, such as the detection of a protein of low abundance, because each protein molecule can be bound by more than one antibody at multiple distinct antigenic sites.
Monoclonal antibodies with virtually any desired specificity can be readily prepared
The discovery of a means of producing monoclonal antibodies of virtually any desired specificity was a major breakthrough that intensified the power of immunological approaches. As with impure proteins, working with an impure mixture of antibodies makes it difficult to interpret data. Ideally, one would isolate a clone of cells producing a single, identical antibody. The problem is that antibody-producing cells isolated from an organism have short life spans.
Immortal cell lines that produce monoclonal antibodies do exist. These cell lines are derived from a type of cancer, multiple myeloma, which is a malignant disorder of antibody-producing cells. In this cancer, a single transformed plasma cell divides uncontrollably, generating a very large number of cells of a single kind. Such a group of cells is a clone because the cells are descended from the same cell and have identical properties. The identical cells of the myeloma secrete large amounts of a single immunoglobulin generation after generation. While these antibodies have proven useful for elucidating antibody structure, nothing is known about their specificity. Hence, they have little utility for the immunological methods described in the next pages.
César Milstein and Georges Köhler discovered that large amounts of antibodies of nearly any desired specificity can be obtained by fusing a short-lived antibody-producing cell with an immortal myeloma cell. An antigen is injected into a mouse, and its spleen is removed several weeks later (Figure 3.20). A mixture of plasma cells from this spleen is fused in vitro with myeloma cells. Each of the resulting hybrid cells, called hybridoma cells, indefinitely produces the identical antibody specified by the parent cell from the spleen. Hybridoma cells can then be screened by a specific assay for the antigen–antibody interaction to determine which ones produce antibodies of the preferred specificity. Collections of cells shown to produce the desired antibody are subdivided and reassayed. This process is repeated until a pure cell line, a clone producing a single antibody, is isolated. These positive cells can be grown in culture medium or injected into mice to induce myelomas. Alternatively, the cells can be frozen and stored for long periods.

FIGURE 3.21Fluorescence micrograph of a developing Drosophila embryo. The embryo was stained with a fluorescence-labeled monoclonal antibody for the DNA-binding protein encoded by engrailed, an essential gene in specifying the body plan.
[Courtesy of Dr. Nipam Patel and Dr. Corey Goodman.]
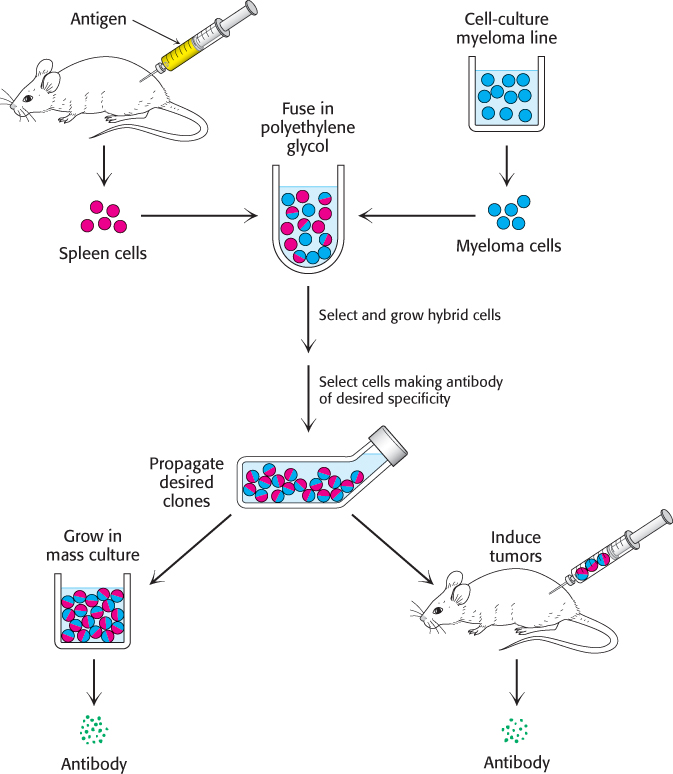
FIGURE 3.20Preparation of monoclonal antibodies. Hybridoma cells are formed by the fusion of antibody-producing cells and myeloma cells. The hybrid cells are allowed to proliferate by growing them in selective medium. They are then screened to determine which ones produce antibody of the desired specificity.
[Information from C. Milstein. Monoclonal antibodies. Copyright © 1980 by Scientific American, Inc. All rights reserved.]
The hybridoma method of producing monoclonal antibodies has opened new vistas in biology and medicine. Large amounts of identical antibodies with tailor-made specificities can be readily prepared. They are sources of insight into relations between antibody structure and specificity. Moreover, monoclonal antibodies can serve as precise analytical and preparative reagents. Proteins that guide development have been identified with the use of monoclonal antibodies as tags (Figure 3.21). Monoclonal antibodies attached to solid supports can be used as affinity columns to purify scarce proteins. This method has been used to purify interferon (an antiviral protein) 5000-fold from a crude mixture. Clinical laboratories use monoclonal antibodies in many assays. For example, the detection in blood of isozymes that are normally localized in the heart points to a myocardial infarction (heart attack). Blood transfusions have been made safer by antibody screening of donor blood for viruses that cause AIDS (acquired immune deficiency syndrome), hepatitis, and other infectious diseases. Monoclonal antibodies can be used as therapeutic agents. For example, trastuzumab (Herceptin) is a monoclonal antibody useful for treating some forms of breast cancer.
Proteins can be detected and quantified by using an enzyme-linked immunosorbent assay
Antibodies can be used as exquisitely specific analytic reagents to quantify the amount of a protein or other antigen present in a biological sample. The enzyme-linked immunosorbent assay (ELISA) makes use of an enzyme that reacts with a colorless substrate to produce a colored product. The enzyme is covalently linked to a specific antibody that recognizes a target antigen. If the antigen is present, the antibody–enzyme complex will bind to it and, on addition of the substrate, the enzyme will catalyze the reaction, generating the colored product. Thus, the presence of the colored product indicates the presence of the antigen. Rapid and convenient, ELISAs can detect less than a nanogram (10−9 g) of a specific protein. ELISAs can be performed with either polyclonal or monoclonal antibodies, but the use of monoclonal antibodies yields more-reliable results.
 We will consider two among the several types of ELISA. The indirect ELISA is used to detect the presence of antibody and is the basis of the test for HIV infection. The HIV test detects the presence of antibodies that recognize viral core protein antigens. Viral core proteins are adsorbed to the bottom of a well. Antibodies from the person being tested are then added to the coated well. Only someone infected with HIV will have antibodies that bind to the antigen. Finally, enzyme-linked antibodies to human antibodies (e.g., enzyme-linked goat antibodies that recognize human antibodies) are allowed to react in the well, and unbound antibodies are removed by washing. Substrate is then applied. An enzyme reaction yielding a colored product suggests that the enzyme-linked antibodies were bound to human antibodies, which in turn implies that the patient has antibodies to the viral antigen (Figure 3.22A). This assay is quantitative: the rate of the color-formation reaction is proportional to the amount of antibody originally present.
We will consider two among the several types of ELISA. The indirect ELISA is used to detect the presence of antibody and is the basis of the test for HIV infection. The HIV test detects the presence of antibodies that recognize viral core protein antigens. Viral core proteins are adsorbed to the bottom of a well. Antibodies from the person being tested are then added to the coated well. Only someone infected with HIV will have antibodies that bind to the antigen. Finally, enzyme-linked antibodies to human antibodies (e.g., enzyme-linked goat antibodies that recognize human antibodies) are allowed to react in the well, and unbound antibodies are removed by washing. Substrate is then applied. An enzyme reaction yielding a colored product suggests that the enzyme-linked antibodies were bound to human antibodies, which in turn implies that the patient has antibodies to the viral antigen (Figure 3.22A). This assay is quantitative: the rate of the color-formation reaction is proportional to the amount of antibody originally present.
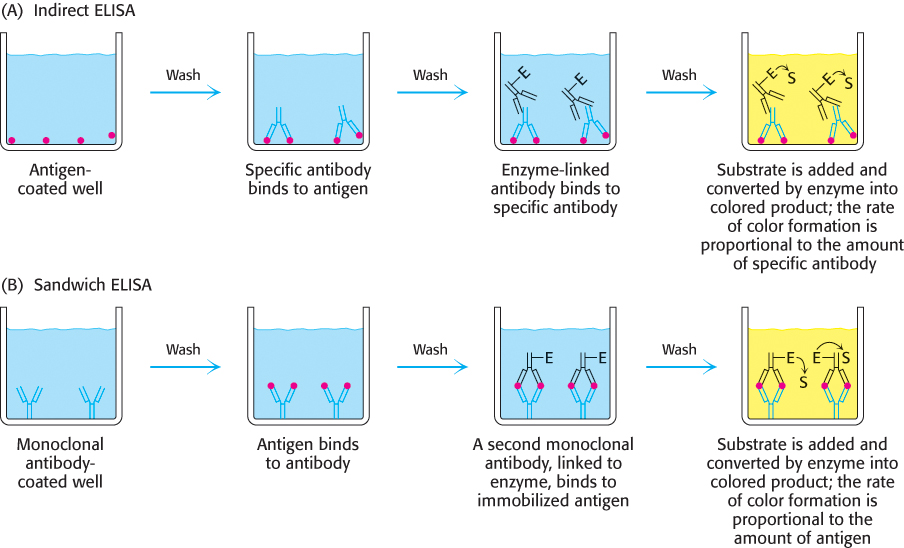
FIGURE 3.22Indirect ELISA and sandwich ELISA. (A) In indirect ELISA, the production of color indicates the amount of an antibody to a specific antigen. (B) In sandwich ELISA, the production of color indicates the quantity of antigen.
[Information from R. A. Goldsby, T. J. Kindt, and B. A. Osborne, Kuby Immunology, 4th ed. (W. H. Freeman and Company, 2000), p. 162.]
The sandwich ELISA is used to detect antigen rather than antibody. Antibody to a particular antigen is first adsorbed to the bottom of a well. Next, solution containing the antigen (such as blood or urine, in medical diagnostic tests) is added to the well and binds to the antibody. Finally, a second, different antibody to the antigen is added. This antibody is enzyme linked and is processed as described for indirect ELISA. In this case, the rate of color formation is directly proportional to the amount of antigen present. Consequently, it permits the measurement of small quantities of antigen (Figure 3.22B).
Western blotting permits the detection of proteins separated by gel electrophoresis
Very small quantities of a protein of interest in a cell or in body fluid can be detected by an immunoassay technique called western blotting (Figure 3.23). A sample is subjected to electrophoresis on an SDS–polyacrylamide gel. A polymer sheet is pressed against the gel, transferring the resolved proteins on the gel to the sheet, which makes the proteins more accessible for reaction. An antibody that is specific for the protein of interest, called the primary antibody, is added to the sheet and reacts with the antigen. The antibody–antigen complex on the sheet can then be detected by rinsing the sheet with a second antibody, called the secondary antibody, that is specific for the primary antibody (e.g., a goat antibody that recognizes mouse antibodies). Typically, the secondary antibody is fused to an enzyme that produces a chemiluminescent or colored product or contains a fluorescent tag, enabling the identification and quantitation of the protein of interest. Western blotting makes it possible to find a protein in a complex mixture, the proverbial needle in a haystack. It is the basis for the test for infection by hepatitis C, where it is used to detect a core protein of the virus. This technique is also very useful in monitoring protein purification and in the cloning of genes.
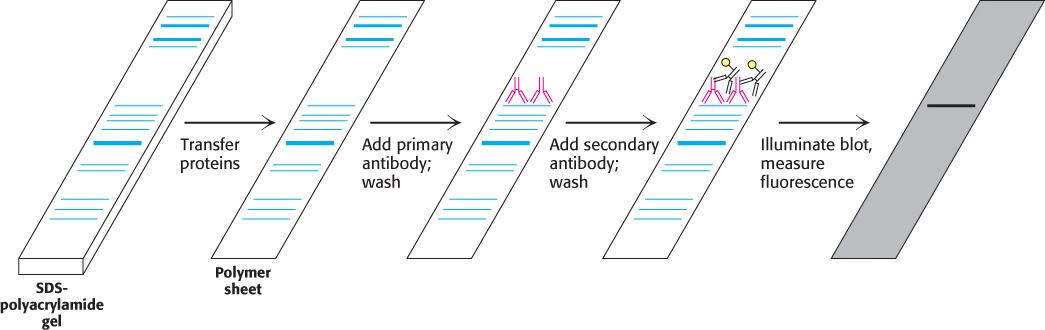
FIGURE 3.23Western blotting. Proteins on an SDS–polyacrylamide gel are transferred to a polymer sheet. The sheet is first treated with a primary antibody, which is specific for the protein of interest, and then washed to remove unbound antibody. Next, the sheet is treated with a secondary antibody, which recognizes the primary antibody, and washed again. Since the secondary antibody is labeled (here, with a fluorescent tag indicated by the yellow circle), the band containing the protein of interest can be identified.
Fluorescent markers make the visualization of proteins in the cell possible
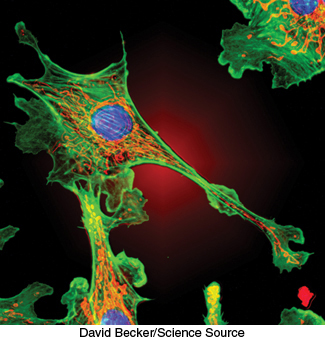
FIGURE 3.24Actin filaments. Fluorescence micrograph of a cell shows actin filaments stained green using an antibody specific to actin.
[David Becker/Science Source.]
Biochemistry is often performed in test tubes or polyacrylamide gels. However, most proteins function in the context of a cell. Fluorescent markers provide a powerful means of examining proteins in their biological context. Cells can be stained with fluorescence-labeled antibodies and examined by fluorescence microscopy to reveal the location of a protein of interest. For example, arrays of parallel bundles are evident in cells stained with antibody specific for actin, a protein that polymerizes into filaments (Figure 3.24). Actin filaments are constituents of the cytoskeleton, the internal scaffolding of cells that controls their shape and movement. By tracking protein location, fluorescent markers also provide clues to protein function. For instance, the mineralocorticoid receptor protein binds to steroid hormones (Section 26.4), including cortisol. The receptor was linked to a yellow variant of green fluorescent protein (GFP), a naturally fluorescent protein isolated from the jellyfish Aequorea victoria (Chapter 2). Fluorescence microscopy revealed that, in the absence of the hormone, the receptor is located in the cytoplasm (Figure 3.25A). On addition of the steroid, the receptor is translocated to the nucleus, where it binds to DNA (Figure 3.25B). These results indicate that the mineralocorticoid receptor protein is a transcription factor that controls gene expression.
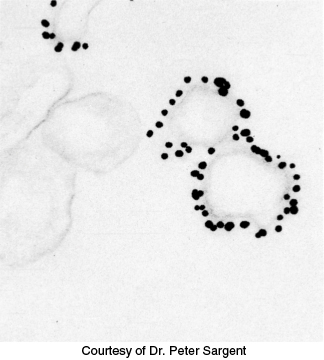
FIGURE 3.26Immunoelectron microscopy. The opaque particles (150-Å, or 15-nm, diameter) in this electron micrograph are clusters of gold atoms bound to antibody molecules. A gold-labeled antibody against a channel protein (Section 13.4) identifies membrane vesicles at the termini of neurons that contain this protein.
[Courtesy of Dr. Peter Sargent.]
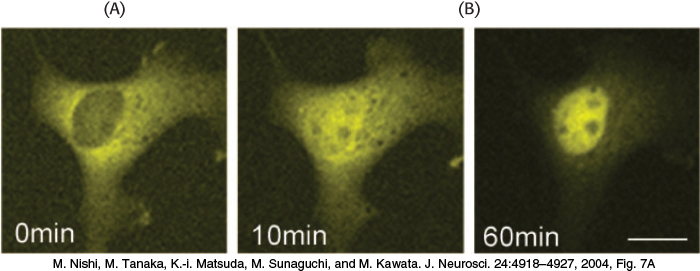
FIGURE 3.25Nuclear localization of a steroid receptor. (A) The mineralocorticoid receptor, made visible by attachment to a yellow variant of GFP, is located predominantly in the cytoplasm of the cultured cell. (B) Subsequent to the addition of corticosterone (a glucocorticoid steroid that also binds to the mineralocorticoid receptor), the receptor moves into the nucleus.
[M. Nishi, M. Tanaka, K.-i. Matsuda, M. Sunaguchi, and M. Kawata. J. Neurosci. 24:4918–4927, 2004, Fig. 7A.]
The highest resolution of fluorescence microscopy is about 0.2 μm (200 nm, or 2000 Å), the wavelength of visible light. Finer spatial resolution can be achieved by electron microscopy if the antibodies are tagged with electron-dense markers. For example, antibodies conjugated to clusters of gold or to ferritin (which has an electron-dense core rich in iron) are highly visible under the electron microscope. Immunoelectron microscopy can define the position of antigens to a resolution of 10 nm (100 Å) or finer (Figure 3.26).





 We will consider two among the several types of ELISA. The indirect ELISA is used to detect the presence of antibody and is the basis of the test for HIV infection. The HIV test detects the presence of antibodies that recognize viral core protein antigens. Viral core proteins are adsorbed to the bottom of a well. Antibodies from the person being tested are then added to the coated well. Only someone infected with HIV will have antibodies that bind to the antigen. Finally, enzyme-
We will consider two among the several types of ELISA. The indirect ELISA is used to detect the presence of antibody and is the basis of the test for HIV infection. The HIV test detects the presence of antibodies that recognize viral core protein antigens. Viral core proteins are adsorbed to the bottom of a well. Antibodies from the person being tested are then added to the coated well. Only someone infected with HIV will have antibodies that bind to the antigen. Finally, enzyme-



