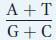7.2 DNA Structure
Even before the structure of DNA was elucidated, genetic studies indicated that the hereditary material must have three key properties:
Because essentially every cell in the body of an organism has the same genetic makeup, faithful replication of the genetic material at every cell division is crucial. Thus, the structural features of DNA must allow faithful replication. These structural features will be considered later in this chapter.
Because it must encode the constellation of proteins expressed by an organism, the genetic material must have informational content. How the information coded in DNA is deciphered to produce proteins will be the subject of Chapters 8 and 9.
Because hereditary changes, called mutations, provide the raw material for evolutionary selection, the genetic material must be able to change on rare occasion. Nevertheless, the structure of DNA must be stable so that organisms can rely on its encoded information. We will consider the mechanisms of mutation in Chapter 16.
DNA structure before Watson and Crick
Consider the discovery of the double-
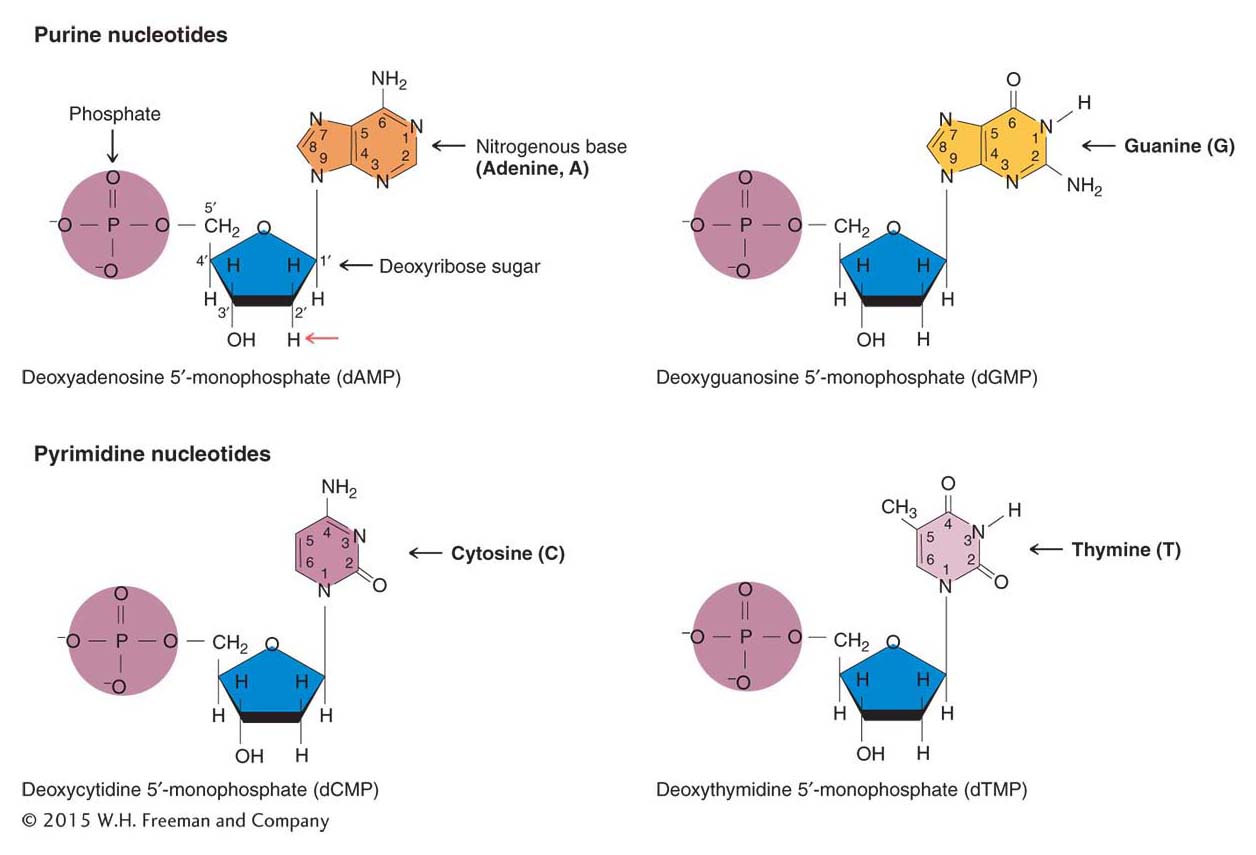
The building blocks of DNA The first piece of the puzzle was knowledge of the basic building blocks of DNA. As a chemical, DNA is quite simple. It contains three types of chemical components: (1) phosphate, (2) a sugar called deoxyribose, and (3) four nitrogenous bases—adenine, guanine, cytosine, and thymine. The sugar in DNA is called “deoxyribose” because it has only a hydrogen atom (H) at the 2′-carbon atom, unlike ribose (a component of RNA), which has a hydroxyl (OH) group at that position. Two of the bases, adenine and guanine, have a double-
The carbon atoms in the bases are assigned numbers for ease of reference. The carbon atoms in the sugar group also are assigned numbers—
The chemical components of DNA are arranged into groups called nucleotides, each composed of a phosphate group, a deoxyribose sugar molecule, and any one of the four bases (Figure 7-5). It is convenient to refer to each nucleotide by the first letter of the name of its base: A, G, C, or T. The nucleotide with the adenine base is called deoxyadenosine 5′-monophosphate, where the 5′ refers to the position of the carbon atom in the sugar ring to which the single (mono) phosphate group is attached.
Chargaff’s rules of base composition The second piece of the puzzle used by Watson and Crick came from work done several years earlier by Erwin Chargaff. Studying a large selection of DNAs from different organisms (Table 7-1), Chargaff established certain empirical rules about the amounts of each type of nucleotide found in DNA:
The total amount of pyrimidine nucleotides (T + C) always equals the total amount of purine nucleotides (A + G).
The amount of T always equals the amount of A, and the amount of C always equals the amount of G. But the amount of A + T is not necessarily equal to the amount of G + C, as can be seen in the right-
hand column of Table 7-1. This ratio varies among different organisms but is virtually the same in different tissues of the same organism.
|
Organism |
Tissue |
Adenine |
Thymine |
Guanine |
Cytosine |
|
|---|---|---|---|---|---|---|
|
Escherichia coli (K12) |
– |
26.0 |
23.9 |
24.9 |
25.2 |
1.00 |
|
Diplococcus pneumoniae |
– |
29.8 |
31.6 |
20.5 |
18.0 |
1.59 |
|
Mycobacterium tuberculosis |
– |
15.1 |
14.6 |
34.9 |
35.4 |
0.42 |
|
Yeast |
– |
31.3 |
32.9 |
18.7 |
17.1 |
1.79 |
|
Paracentrotus lividus (sea urchin) |
Sperm |
32.8 |
32.1 |
17.7 |
18.4 |
1.85 |
|
Herring |
Sperm |
278 |
27.5 |
22.2 |
22.6 |
1.23 |
|
Rat |
Bone marrow |
28.6 |
28.4 |
21.4 |
21.5 |
1.33 |
|
Human |
Thymus |
30.9 |
29.4 |
19.9 |
19.8 |
1.52 |
|
Human |
Liver |
30.3 |
30.3 |
19.5 |
19.9 |
1.53 |
|
Human |
Sperm |
30.7 |
31.2 |
19.3 |
18.8 |
1.62 |
|
* Defined as moles of nitrogenous constituents per 100 g- Source: Data from E. Chargaff and J. Davidson, eds., The Nucleic Acids. Academic Press, 1955. |
||||||
X-

The double helix
A 1953 paper by Watson and Crick in the journal Nature began with two sentences that ushered in a new age of biology: “We wish to suggest a structure for the salt of deoxyribose nucleic acid (D.N.A.). This structure has novel features which are of considerable biological interest.”1 The structure of DNA had been a subject of great debate since the experiments of Avery and co-

The three-
Each base is attached to the 1′-carbon atom of a deoxyribose sugar in the backbone of each strand and faces inward toward a base on the other strand. Hydrogen bonds between pairs of bases hold the two strands of the DNA molecule together. The hydrogen bonds are indicated by dashed lines in Figure 7-8b.
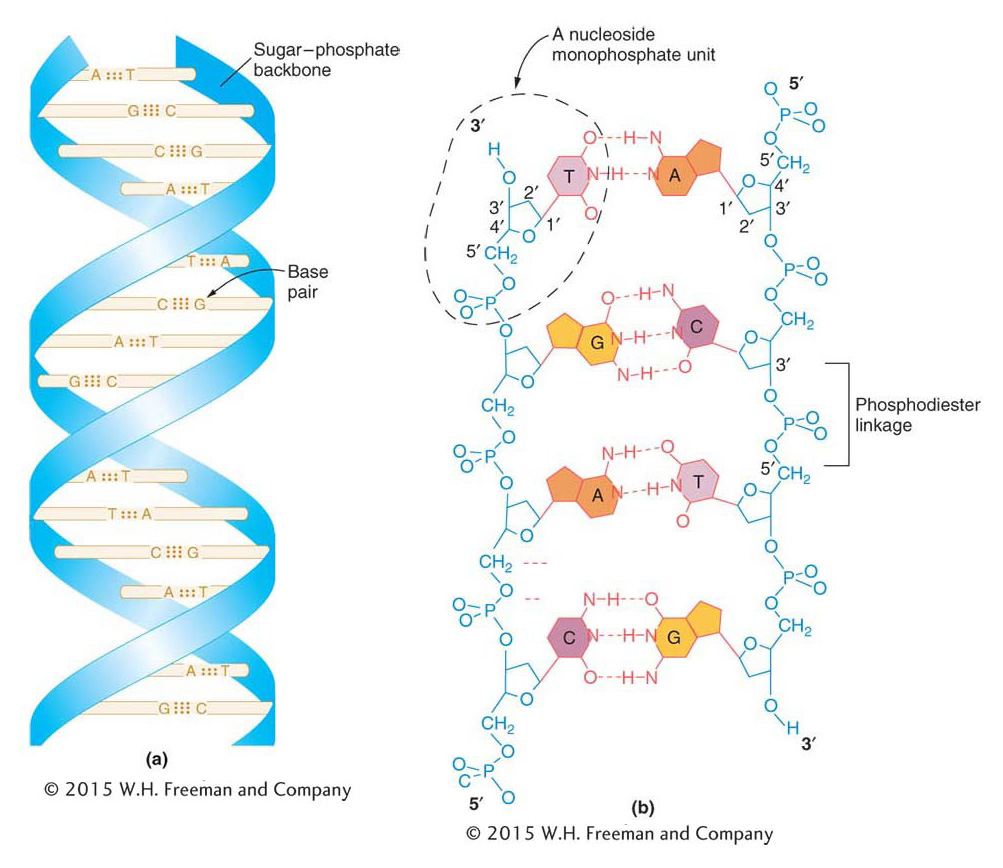
Two complementary nucleotide strands paired in an antiparallel manner automatically assume a double-
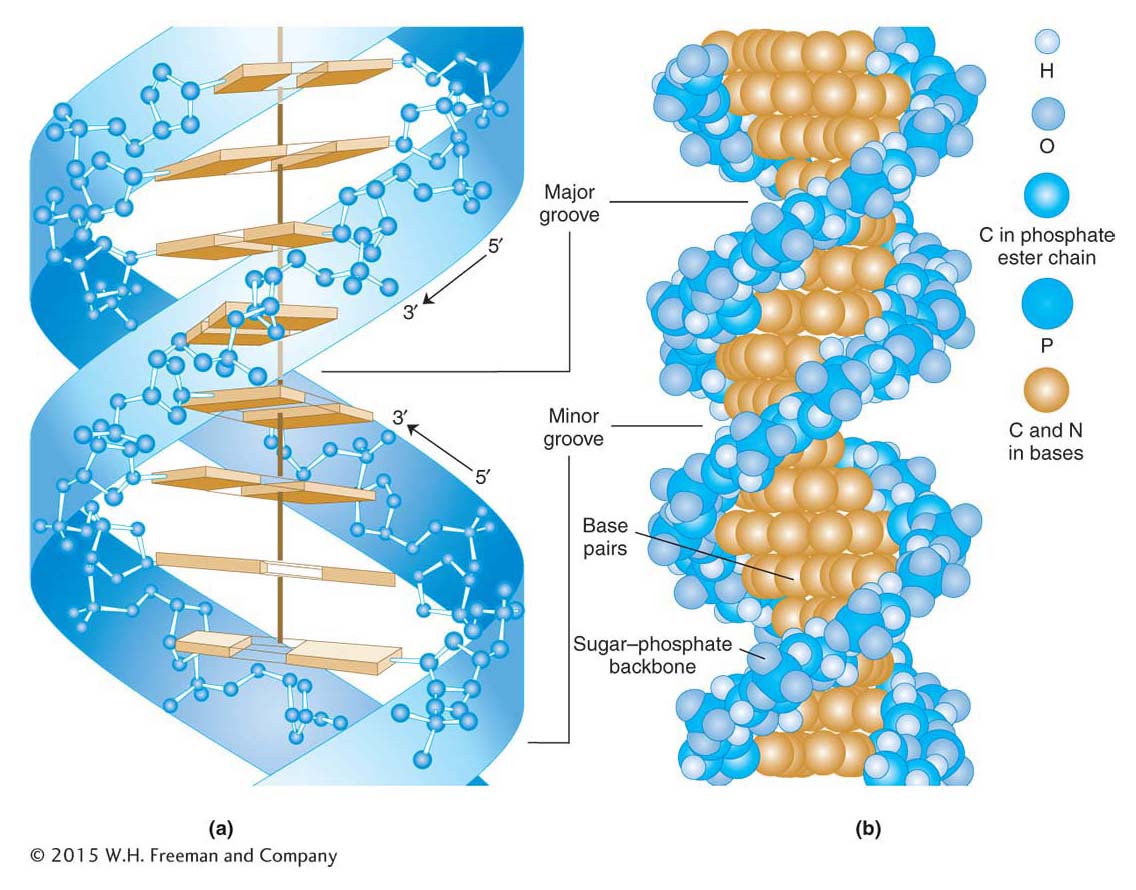
The double helix accounted nicely for the X-
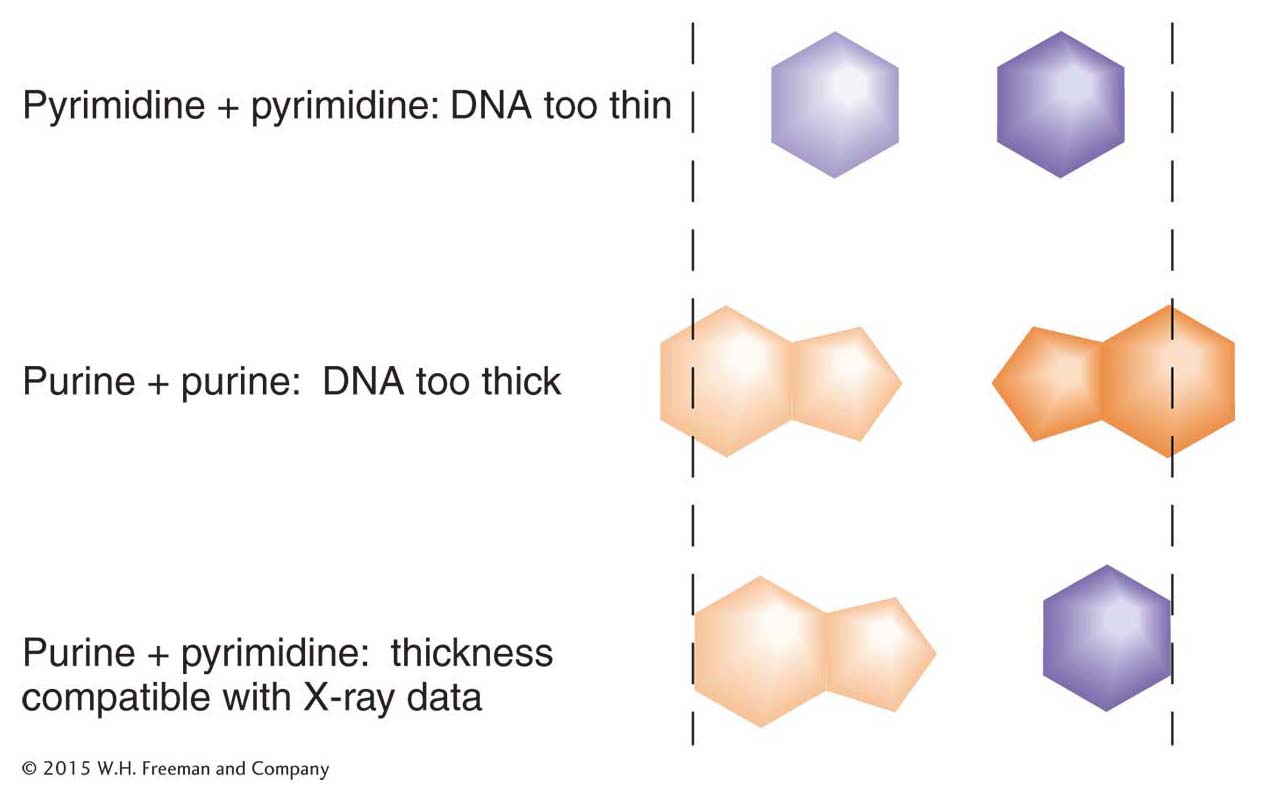
Note that the G–
KEY CONCEPT
DNA is a double helix composed of two nucleotide chains held together by complementary pairing of A with T and G with C.Watson and Crick’s discovery of the structure of DNA is considered by some to be the most important biological discovery of the twentieth century and led to their being awarded the Nobel Prize with Maurice Wilkins in 1962 (Rosalind Franklin died of cancer in 1958 and the prize is not awarded posthumously). The reason that this discovery is considered so important is that the double helix model, in addition to being consistent with earlier data about DNA structure, fulfilled the three requirements for a hereditary substance:
The double-
helical structure suggested how the genetic material might determine the structure of proteins. Perhaps the sequence of nucleotide pairs in DNA dictates the sequence of amino acids in the protein specified by that gene. In other words, some sort of genetic code may write information in DNA as a sequence of nucleotides and then translate it into a different language of amino acid sequences in protein. Just how it is done is the subject of Chapter 9. If the base sequence of DNA specifies the amino acid sequence, then mutation is possible by the substitution of one type of base for another at one or more positions. Mutations will be discussed in Chapter 16.
As Watson and Crick stated in the concluding words of their 1953 Nature paper that reported the double-
helical structure of DNA: “It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material.”2 To geneticists at the time, the meaning of this statement was clear, as we see in the next section.