CONCEPT38.2 Cleavage Creates Building Blocks and Produces a Blastula
Cleavage is the initial process that creates cellular building blocks to form the new organism by producing many smaller cells from one. Ultimately, as a result of cleavage, these smaller cells will differ in their cytoplasmic determinants. To understand cleavage, it is important to understand that no growth occurs at this stage. Cleavage divisions differ from other mitotic divisions in that there is no “gap” or growth phase between divisions, so no new cytoplasm is added. Thus as cells increase in number, they become smaller and smaller. The cells produced are called blastomeres. The cluster of cells produced is termed a blastula.
Early investigators thought that, through cleavage, genes were divided up so that each blastomere would later form its specific tissue or organ according to its restricted genetic makeup. We now know that this is not the case. Each blastomere receives the entire genome of the animal. Now we have the next mystery: How can cells that all have the same genetic information give rise to the diversity of cell types that we see in an adult? The answer depends on several mechanisms (see Concept 14.2).
During early stages of development, events are controlled by specific information passed on by the mother. Proteins and mRNAs, referred to as cytoplasmic determinants, are deposited in the cytoplasm of the egg as it matures. Early cleavages occur rapidly, and there is little or no transcription of mRNA in the zygote nucleus. Rather, mRNA already placed by the mother in the egg is used to direct protein synthesis. In most animals, it is not until the mid- to late blastula stage that the genome of the developing embryo takes over. Prior to that time, maternal factors control development. As we saw in the formation of the gray crescent, the cytoskeleton may redistribute such materials from one point to another. Also, at some point, as mitotic divisions continue, new cell membranes separate cytoplasmic determinants so they become distributed differentially among blastomeres (see Concept 14.2, especially Figure 14.7). These processes plus cell–cell communication play a role in the determination of blastomere fates. All of these processes are involved in the development of most animals, but as you will see, the stage at which each process comes into play differs from group to group. At some point, the fate of a blastomere becomes restricted or determined and it can no longer form an entire embryo.
Scientists have learned much about the developmental potential of blastomeres by separating them at different times of development. In such experiments, blastomeres are isolated to learn how they will develop on their own. It is also possible to leave all the cells together, label a specific cell (e.g., with a dye), and then identify the tissues and organs that form from that cell. A new technique called light sheet microscopy allows individual cells to be followed through many cleavages without the addition of a dye. Experiments of all types—isolation experiments and tracing experiments—have been used to generate fate maps for many species (FIGURE 38.3). To understand this, we need to explore different cleavage patterns.
Cleavage is easiest to understand in animals that produce eggs with little yolk, such as sea urchins. Blastomeres may be the same size or different. They will be the same size if the egg cytoplasm is divided equally among all the blastomeres. Unequal cytoplasmic divisions will result in large blastomeres and small blastomeres. As cleavage continues, the zygote becomes divided into progressively smaller blastomeres:
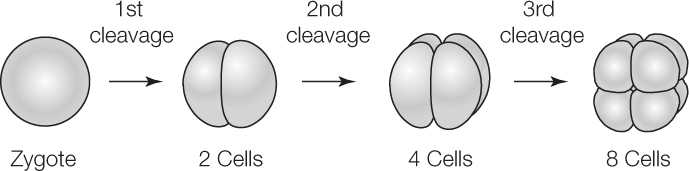
If the cytoplasm is completely divided by each cell division, cleavage is termed complete cleavage. If it is not, as you will see is the case in birds, insects, and some other animals, it is termed incomplete cleavage.
Two different patterns of cleavage that seem to have deep evolutionary roots are radial cleavage and spiral cleavage. These patterns are seen in eggs that have little yolk and undergo complete cleavage, such as those of snails and sea urchins. In all types of cleavage, the plane of cleavage (shown by the position of the new cell membrane that divides the cells) is determined by the orientation of the mitotic spindle. Differences in radial and spiral cleavage are readily seen after the third cleavage (the 8-cell stage).
At this time, in radial cleavage the four daughter cells lie directly above the four parent cells:
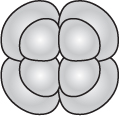
791
During spiral cleavage, mitotic spindles align so that, after the third and following cleavages, the four daughter cells lie in the grooves between the four parent cells, creating the appearance of a spiral:
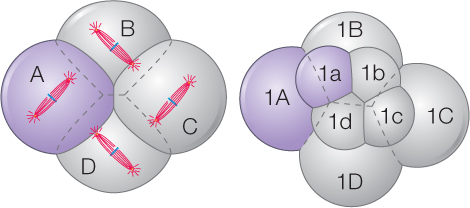
A spirally cleaving egg has the most thermodynamically stable shape possible. Appropriately sized soap bubbles assume the same configuration as a spirally cleaving egg.
You are probably most familiar with deuterostome animals, the large clade (group) that includes the vertebrates and all other chordates, echinoderms, and a few other groups. Echinoderms and several other deuterostomes display radial cleavage. Radial cleavage is considered to be the basic deuterostome pattern, although it is often highly modified in the presence of yolk.
The other large group of animals, the protostomes, includes most of the invertebrates. Protostomes do not have radial cleavage. Here we focus on the lophotrochozoans, a clade that includes about half of all animal phyla, including the annelids and mollusks. In the lophotrochozoans, spiral cleavage predominates.
LINK
Concept 23.1 shows how early development determines body plans that reflect phylogenetic relationships among the animals; Concept 23.3 discusses the protostomes, explaining that there are two major clades; Concept 23.5 discusses the deuterostomes and some of their developmental characteristics
Specific blastomeres generate specific tissues and organs
Blastomeres become determined—irreversibly committed to specific fates—at different times in different species. In many protostomes with spirally cleaving eggs and in a few deuterostomes, blastomere fates are determined as early as the 2-cell stage. If one of these blastomeres is experimentally removed, a particular portion of the embryo will not form. This type of development has been called mosaic development because each blastomere appears to contribute a specific set of “tiles” to the final “mosaic” that is the adult animal (FIGURE 38.4).
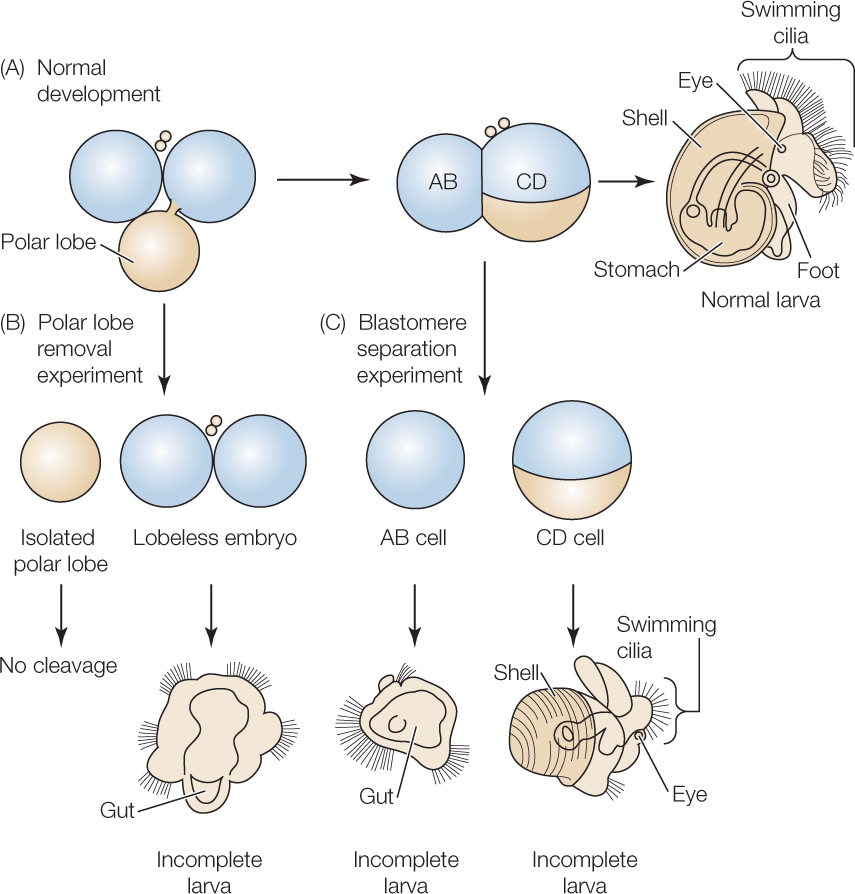
A feature of many spirally cleaving eggs that is not seen in radially cleaving eggs is the production of a polar lobe, at the vegatal pole, during early cleavages. Cytoplasmic determinants as well as yolk are sequestered in this “out-pouching” during cell division and are then returned to a specific blastomere after cell membranes have re-formed. If the polar lobe is removed experimentally, one abnormal larva develops that typically lacks specific structures (see Figure 38.4B). If the two blastomeres are experimentally separated, two abnormal larvae develop, each lacking very specific structures (see Figure 38.4C). These experiments clearly demonstrate one way in which information for further development is distributed to specific blastomeres.
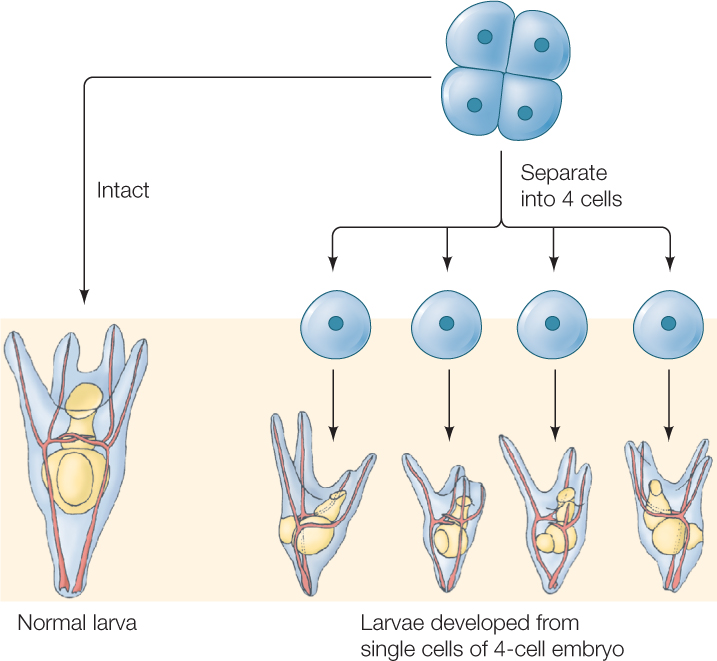
In contrast to mosaic development, in regulative development the loss of some cells during cleavage does not affect the developing embryo. Instead, the remaining cells compensate for the loss. If blastomeres of a sea urchin egg are separated at the 2-cell stage, two half-sized, but complete, larvae form. Even at the 4-cell stage, separation produces four small larvae (FIGURE 38.5). We now know that cytoplasmic determinants are found in the cytoplasm of eggs showing both mosaic and regulative development. In animals that display regulative development, however, the distribution of these determinants happens later as cell divisions continue (see Concept 14.2; Figure 14.7). Regulative development is typical of vertebrate species, including humans.
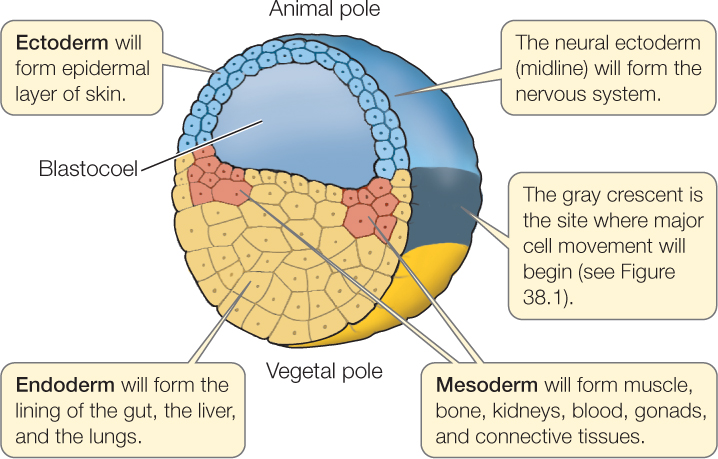
As noted earlier, cleavage leads to the production of a ball of cells termed a blastula. Blastulas produced by radially cleaving eggs, and by those whose cleavage is derived from this basic pattern, have an inner fluid-filled cavity called a blastocoel (see Figure 38.3). The blastula of a sea urchin, for example, is a hollow sphere. Blastulas produced by spirally cleaving eggs may have a blastocoel, but often they are a solid ball of cells, without the fluid-filled space.
792
The amount of yolk affects cleavage
The amount of yolk has a profound effect on cleavage patterns and other developmental processes such as gastrulation, which we will discuss in Concept 38.3. Some eggs, such as those of sea urchins, snails, and placental mammals, have little yolk. Some, such as those of frogs and salamanders, have a moderate amount of yolk. Others, including those of birds and egg-laying mammals, are extremely yolky. As you will see, it is as if the presence of yolk slows down or impedes cell divisions. This means that the eggs of many animals do not follow the radial or spiral cleavage patterns that we consider basic in deuterostomes and protostomes.
In eggs with little yolk, like those of sea urchins and snails, cleavage usually proceeds as described above and is complete.
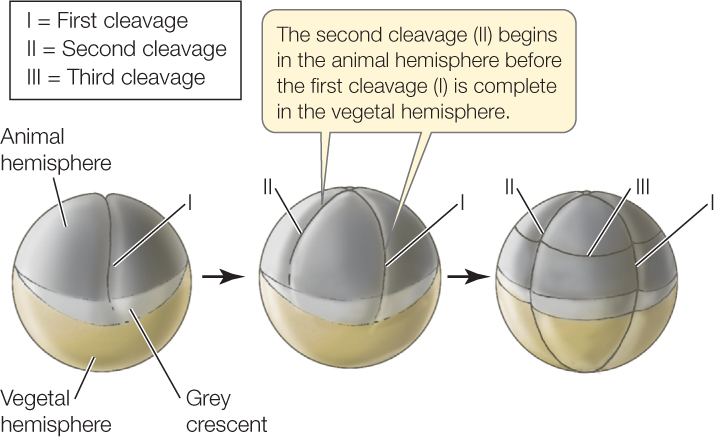
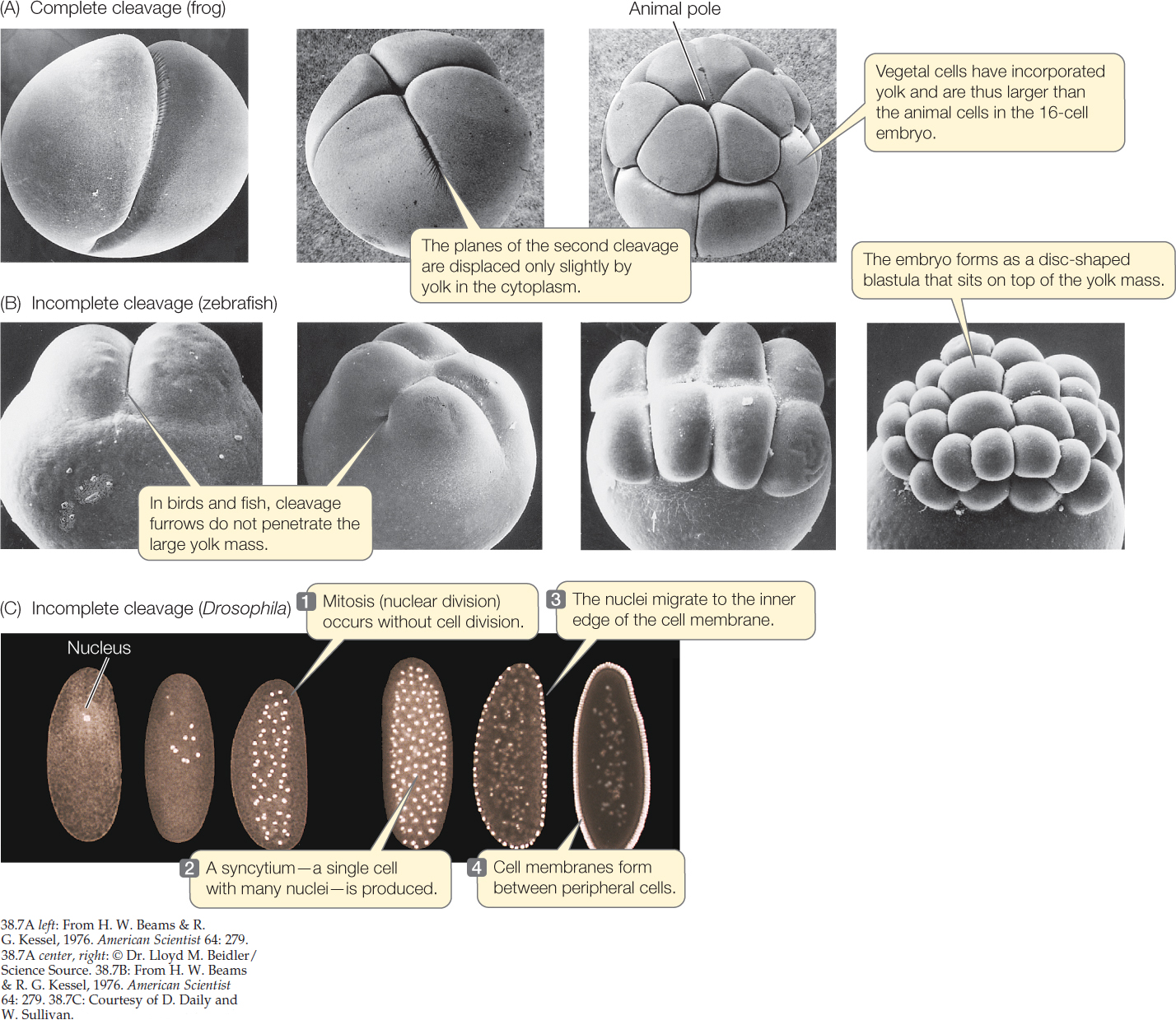
In moderately yolky eggs like those of a frog, cleavage is complete and cell membranes form around every nucleus. However, cells divide more slowly in the yolk-filled vegetal hemisphere than in the animal hemisphere (FIGURES 38.6 and 38.7A). In the vegetal hemisphere, the yolk retards the process of division so that the second round of cleavage has begun in the animal hemisphere before the first round is completed in the vegetal hemisphere. In the example shown in Figure 38.6, the third cleavage is horizontal and occurs in the animal hemisphere. Because cells divide faster in the animal hemisphere, more cells are produced in the dorsal portion of the egg than in the ventral, resulting in an animal hemisphere consisting of small blastomeres and a vegetal hemisphere consisting of fewer, larger blastomeres. The blastocoel forms within the small blastomeres of the animal hemisphere.
In very yolky eggs such as those of a squid (a molluscan protostome), fish, bird, or egg-laying mammal (e.g., the platypus), the yolk remains undivided and the embryo forms as a small blastodisc of cleaving cells on the yolk surface (FIGURE 38.7B). The yolk never divides, and so the cleavage is incomplete. The disc of cells on top of the yolk enlarges and becomes several cells thick. For example, the blastula of a bird, or of the zebrafish shown in Figure 38.7B, is a flattened structure that sits atop the large undivided yolk and consists of an upper layer called epiblast, which will give rise to the embryo proper, and a lower hypoblast, with a fluid-filled space between the two. (Epiblast and hypoblast in the bird egg are shown in Figure 38.11.)
A different type of incomplete cleavage is seen in insects. In Drosophila eggs, for example, the original zygote nucleus undergoes a series of divisions unaccompanied by divisions of the cytoplasm. This early embryo, then, is a syncytium: a mass of cytoplasm in which several nuclei are surrounded by the same cell membrane. As development proceeds, nuclei migrate to the periphery of the egg, and cell membranes form (FIGURE 38.7C).
Cleavage in placental mammals is unique
Placental mammals have non-yolky eggs (which undergo development within the mother’s body after fertilization), but their cleavage pattern differs from that of other non-yolky deuterostome eggs. You might expect that their eggs would show radial cleavage like sea urchin eggs. They do not. Remember that mammals evolved from a reptilian ancestor. As a result, several aspects of their development—such as the formation of epiblast and hypoblast, as well as of the yolk sac and allantois (which we will discuss in Concept 38.5)—were derived from processes in yolky eggs of reptiles, including birds. Other features, however, are unique. The two groups of placental mammals—the marsupials and the eutherians (see Concept 23.6) have evolved slightly different developmental steps. In this chapter, we will consider only eutherian development. Early cell divisions in eutherians do not follow the radial cleavage pattern. For example, at the second cleavage division, the two blastomeres divide in different planes, so that one blastomere cleaves vertically and the other horizontally, in a pattern called rotational cleavage:
793
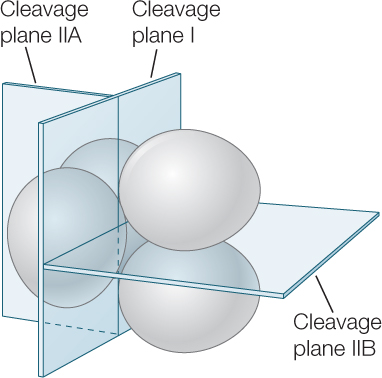
Blastomeres within the same egg divide at different times, so the eutherian egg may contain an odd number of cells. This is not usually the case during early cleavage in other groups. Finally, the mammalian genome is activated very early, so cleavage and development are governed by the new nuclei rather than by cytoplasmic determinants passed on from the mother. In most deuterostomes, this transition happens toward the end of the blastula stage.
Another unique feature of mammalian development occurs during the fourth cleavage, when the cells separate into two groups (FIGURE 38.8A). The inner cell mass will become the embryo, while the surrounding outer cells become an encompassing sac called the trophoblast. Trophoblast cells secrete fluid, creating a blastocoel with the inner cell mass at one end. At this stage, the mammalian embryo is called a blastocyst. Later, trophoblast cells will contribute to the formation of a unique mammalian organ, the placenta, which we will discuss in Concept 38.5.
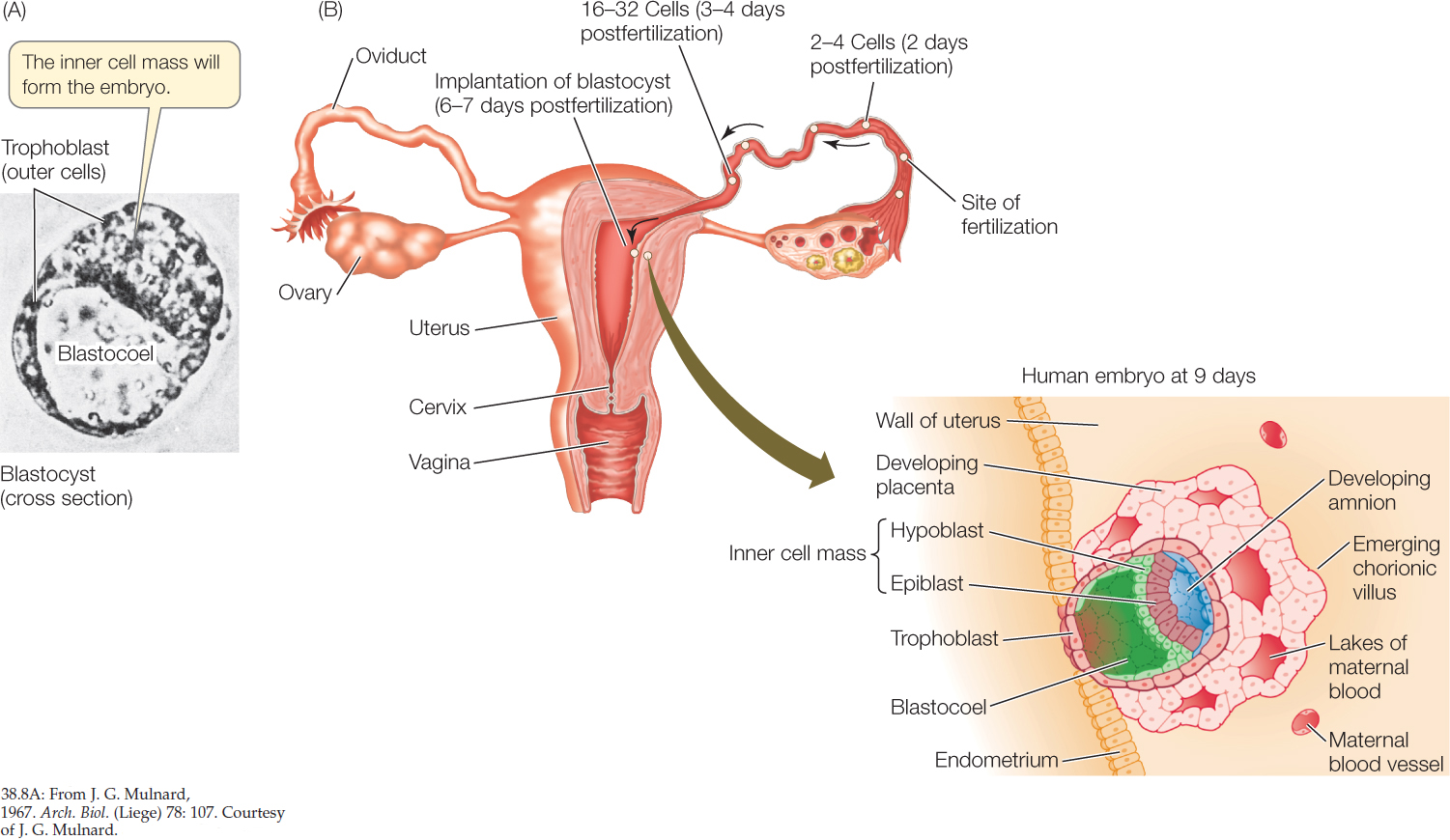
In a mammal, fertilization occurs in the upper reaches of the mother’s oviduct, and cleavage takes place as the zygote travels down the oviduct. When the blastocyst arrives in the uterus, it adheres to the lining of the uterus, which is called the endometrium. This starts the process of implantation in which the early embryo burrows into the uterine wall. In humans, implantation begins about 6 days after fertilization (FIGURE 38.8B).
794
CHECKpointCONCEPT38.2
- What two major functions does cleavage perform?
- Compare radial and spiral cleavage. What determines the direction of the cleavage plane?
- What does experimental removal of the polar lobe of a snail egg demonstrate?
- Describe complete and incomplete cleavage and explain where mammals fit into this scheme.
- What type of development must a species have for identical twins to occur? What do you think leads to the development of such twins? How do you think fraternal twins are produced?
After formation of the blastula, the next stage of development is gastrulation. The developmental biologist Louis Wolpert once said, “It is not birth, marriage, or death, but gastrulation which is the most important time in your life.”