7-5 Chemical and Genetic Measures of Brain and Behavior
Section 3-2 investigates how neurons function. Section 3-3 relates genes to cell function, genetic engineering, and epigenetic mechanisms.
Our focus so far has been on how neuroscientists study the individual and collective activity of neurons and how neuronal activity relates to behavior. Neurons are regulated by genes, DNA segments that encode the synthesis of particular proteins within cells. Genes control the cell’s production of chemicals, so it is possible to relate behavior to genes and to chemicals inside and outside the cell. Chemical and genetic approaches require sophisticated technologies that have seen major advances in the past decade.
Measuring Brain Chemistry
The brain contains a wide mixture of chemicals ranging from neurotransmitters and hormones to glucose and gases, among many others. Abnormalities in amounts of these chemicals can cause serious disruptions in behavior. Prime examples are Parkinson disease, characterized by low dopamine levels in the substantia nigra, and depression, correlated with low serotonin and/or noradrenaline production. The simplest way to measure brain chemistry in such diseases is to extract tissue postmortem from affected humans or laboratory animals and undertake traditional biochemical techniques, such as high-
Section 12-6 explores neural effects of rewarding events.
Fluctuations in brain chemistry are associated not only with behavioral dysfunction but also with ongoing healthy behavior. For example, research over at least the past 30 years shows that dopamine levels fluctuate in the nucleus accumbens (a structure in the subcortical basal ganglia) in association with stimuli related to rewarding behaviors such as food and sex. Changes in brain chemistry can be measured in freely moving animals using two methods, cerebral microdialysis and cerebral voltammetry.
Section 4-1 explains diffusion and concentration gradients in detail.
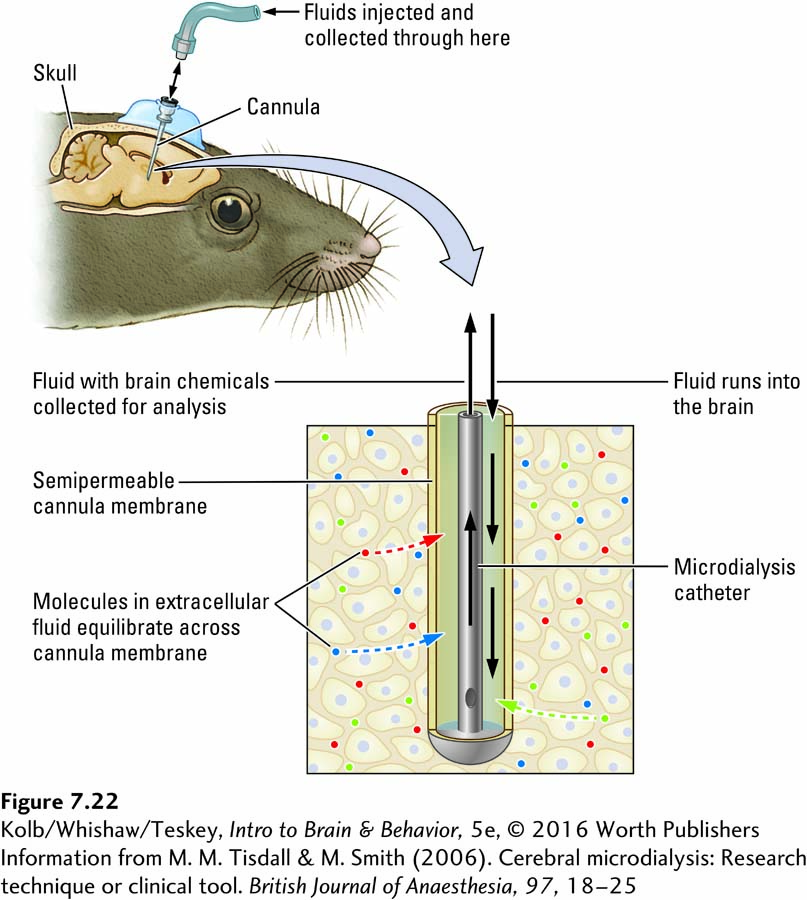
Microdialysis, which can determine the chemical constituents of extracellular fluid, is widely used in the laboratory. The technique has found clinical application over the past decade. A catheter with a semipermeable membrane at its tip is placed in the brain, as illustrated in Figure 7-22. A fluid flows through the cannula and passes along the cell membrane. Simple diffusion drives extracellular molecules across the membrane along their concentration gradient.
This result mirrors wanting-
Fluid containing the molecules from the brain exits through tubing to be collected for analysis. The fluid is removed at a constant rate so that changes in brain chemistry can be correlated with behavior. For example, if a rat is placed in an environment in which it anticipates sex or a favored food, microdialysis will record an increase in dopamine within the basal ganglia regions of the caudate nucleus and putamen, known as the striatum.
Section 6-4 investigates why glutamate and similar chemicals can act as neurotoxins.
Microdialysis is used in some medical centers to monitor chemistry in the injured brain. The effects of TBI or stroke can be worsened by secondary events such as a drastic increase in the neurotransmitter glutamate. Such biochemical changes can lead to irreversible cell damage or death. Physicians are beginning to use microdialysis to monitor such changes, which can then be treated.
Cerebral voltammetry works on a different principle. A small carbon fiber electrode and a metal electrode are implanted in the brain, and a weak current is passed through the metal electrode. The current causes electrons to be added to or removed from the surrounding chemicals. Changes in extracellular levels of specific neurotransmitters can be measured as they occur.
Because different currents lead to changes in different compounds, it is possible to identify levels of different transmitters, such as serotonin or dopamine, and related chemicals. Voltammetry has the advantage of not requiring the chemical analysis of fluid removed from the brain, as microdialysis does, but it has the disadvantage of being destructive. That is, the chemical measurements require the degradation of one chemical into another. Thus this technique is not well suited to clinical uses.
Measuring Genes in Brain and Behavior
Most human behaviors cannot be explained by genetic inheritance alone, but variations in gene sequences do contribute significantly to brain organization. About 1 in 250 live births are identical twins, people who share an identical genome. Identical twins often have remarkably similar behavioral traits. Twin studies show strong concordance rates that support genetic contributions to drug addiction and other psychiatric disorders. But twin studies also show that environmental factors and life experience must be involved: concordance for most behavioral disorders, such as schizophrenia and depression, is far less than 100 percent. Life experiences are acting epigenetically to alter gene expression.
Section 8-2 explains how neurotrophic factors, nourishing chemical compounds, support growth and differentiation in developing neurons.
Genetic factors can also be studied by comparing people who were adopted early in life and usually would not have a close genetic relationship to their adoptive parents. Here, a high concordance rate for behavioral traits would imply a strong environmental influence on behavior. Ideally, an investigator would be able to study both the adoptive and biological parents to tease out the relative heritability of behavioral traits.
7-3
Cannabis Use, Psychosis, and Genetics
Cannabis is the most widely used illicit drug in the world. Because it is nearly impossible to overdose, cannabis is usually considered safe, but it is not completely free of side effects. A relationship has been established between cannabis use and the emergence of psychosis, especially with use in adolescence: earlier use is associated with both an earlier onset and worse long-
Given that most adolescents who use cannabis do not develop psychosis, however, some genetic vulnerability likely predisposes certain individuals to develop a psychotic condition when exposed to it. Cannabis use thus may act more as a trigger for the onset of schizophrenia than as the primary cause of the disease.
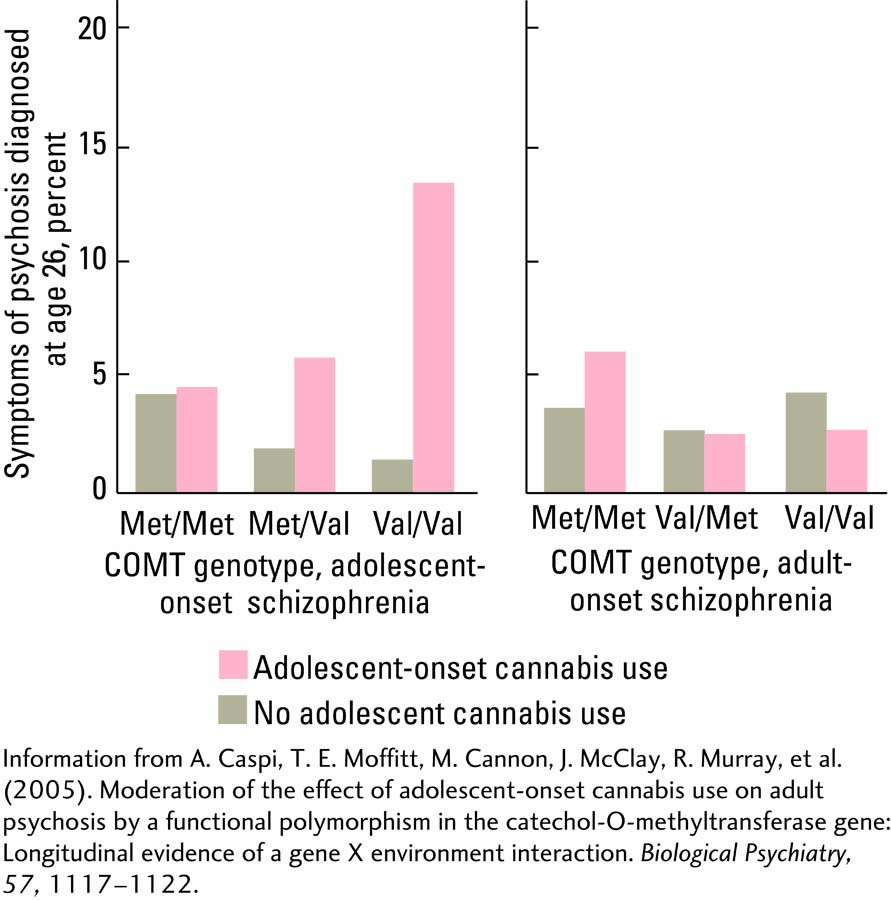
A working hypothesis contends that one culprit may be the COMT gene, because COMT has been associated with schizophrenia. The COMT gene product is an enzyme involved in metabolizing dopamine in the synapse, and abnormalities in dopaminergic activity are associated with psychosis. The hypothesis predicts that adolescents who develop psychosis after cannabis use have an abnormality in the COMT gene.
Avshalom Caspi and colleagues (2005) analyzed the COMT gene in nearly a thousand 26-
These results show that genetic variations can predispose people to show adverse effects of environmental experiences and that the experiences may have age-
Sections 2-6 and 5-3 introduce factors that contribute to dementias. Focus 14-3 describes research and Section 16-3, treatments.
With the development of relatively inexpensive methods of identifying specific genes in people, it is now possible to relate the alleles (different forms) of specific genes to behaviors. A gene related to the production of a compound called brain-
Focus 6-4 relates a case in which chronic cannabis use may have led to psychosis.
Joshua Bueller and his colleagues (2006) showed that the Met allele is associated with an 11 percent reduction in hippocampal volume in healthy participants. Other studies have associated the Met allele with poor memory for specific events (episodic memory) and a high incidence of dementia later in life. However, the Val allele is by no means the better variant: although Val carriers have better episodic memory, they also have a higher incidence of neuroticism and anxiety disorders, as illustrated in Clinical Focus 7-3, Cannabis Use, Psychosis, and Genetics. The two alleles produce different phenotypes because they influence brain structure and functions differently. Other genes that were not measured also differed among Bueller’s participants and may have contributed to the observed difference.
Epigenetics: Measuring Gene Expression
See A Case of Inheriting Experience in Section 3-3.
An individual’s genotype exists in an environmental context fundamental to gene expression, the way genes become active or not. While epigenetic factors do not change the DNA sequence, the genes that are expressed can change dramatically in response to environment and experience. Epigenetic changes can persist throughout a lifetime and even across multiple generations.
Changes in gene expression can result from widely ranging experiences, including chronic stress, traumatic events, drugs, culture, and disease. A study by Mario Fraga and his colleagues (2005) stands as a powerful example of gene–
Although twins’ patterns of gene expression were virtually identical when measured in childhood, 50-
The role of epigenetic differences can also be seen across populations. Moshe Szyf, Michael Meaney, and their colleagues (e.g., 2008) have shown, for example, that the amount of maternal attention mother rats give to their newborn pups alters the expression of certain genes in the adult hippocampus. These genes are related to the infants’ stress response when they are adults. (Maternal attention is measured as the amount and type of mother–
A subsequent study by the same group (McGowan et al., 2009) examined epigenetic differences in hippocampal tissue obtained from two groups of humans: (1) suicides with histories of childhood abuse and (2) either suicides with no childhood abuse or controls who died of other causes. The epigenetic changes found in the abused suicide victims parallel those found in the rats with inattentive mothers, again suggesting that early experiences can alter hippocampal organization and function via changes in gene expression.
Experience-
Consult the Index entry epigenetics to locate coverage throughout the book.
Epigenetic studies promise to revolutionize our understanding of gene–
7-5 REVIEW
Chemical and Genetic Measures of Brain and Behavior
Before you continue, check your understanding.
Question 1
Concentrations of different chemicals in the brain can be measured in postmortem tissue using a(n) ________ assay or in vivo using ________ or ________.
Question 2
Gene–
Question 3
The study of genes and behavior focuses on individual differences in ________, whereas the study of epigenetics and behavior examines differences in ________.
Question 4
Describe briefly how epigenetic studies have led to the recognition that life experience and the environment can alter brain function.
Answers appear in the Self Test section of the book.