14-4 Structural Basis of Brain Plasticity
We have encountered three categories of memory—
Cajal’s neuron theory—
Researchers still encounter a major challenge as they investigate Cajal’s suggestion because where in the brain to look for synaptic changes that might correlate with memory for a specific stimulus is unclear. The task is formidable. Imagine trying to find the exact location of the neurons responsible for storing your grandmother’s name. You would face a similar challenge in trying to pinpoint the neurons responsible for the memory of an object in a monkey’s brain as the monkey performs the visual recognition task illustrated in Figure 14-10B.
One approach to finding memory’s neuronal correlates aims first to determine that synaptic changes do correlate with memory in the mammalian brain; second, to localize the synaptic changes to specific neural pathways; and third, to analyze the synaptic changes themselves. This section reviews studies that begin to show how experience correlates with synaptic changes related to memory. We first consider a strategy based on neuronal physiology and experience. We then look at gross neural changes that correlate with select experiences. These range from potentially good—
Long-Term Potentiation
Experiment 5-2 explains habituation at the neuronal level, Experiment 5-3, sensitization.
Findings from studies of behavioral habituation (a weakened response to a stimulus) and sensitization (a strengthened response) in the sea slug Aplysia show that physical changes in synapses do underlie learning. Adaptive synapses in the mammalian brain participate in associative learning, a response elicited by linking unrelated stimuli together—
Learned associations are a common type of explicit memory. Associating a face with a person, an odor with a food, a sound with a musical instrument are everyday examples. Learning that learning takes place at synapses is another. The phenomenon underlying associative learning entails an enduring neural change in a postsynaptic cell after an excitatory signal, or EPSP, from the presynaptic cell crosses the synaptic gap.
Postsynaptic potentials increase (excitatory/EPSP) or decrease (inhibitory/IPSP) the probability that an action potential will occur. See Experiment 4-1.
Both the relatively simple circuitry of the hippocampus and the ease of recording postsynaptic potentials there make it ideal for studying the neural basis of associative learning. In 1973, Timothy Bliss and Terje Lømø demonstrated that repeated electrical stimulation of the pathway entering the hippocampus progressively increases EPSP size as recorded from hippocampal cells. The enhancement in the size of these graded field potentials lasts several hours to weeks or even longer. Bliss and Lømø called it long-
Deep brain stimulation used for severely depressed subjects induces a change, similar to LTP, which appears to increase brain plasticity; see Section 16-1.
Figure 14-17A illustrates the experimental procedure for obtaining LTP. The presynaptic neuron is stimulated electrically while the electrical activity the stimulation produces is recorded from the postsynaptic neuron. The readout in Figure 14-17A shows the EPSP produced by a single pulse of electrical stimulation. In a typical experiment, many test stimuli are given to estimate the size of the induced EPSP. Then a strong burst of stimulation, consisting of a few hundred pulses of electrical current per second, is administered (Figure 14-17B). The test pulse is then given again. The increased amplitude of the EPSP endures for as long as 90 minutes after the high-
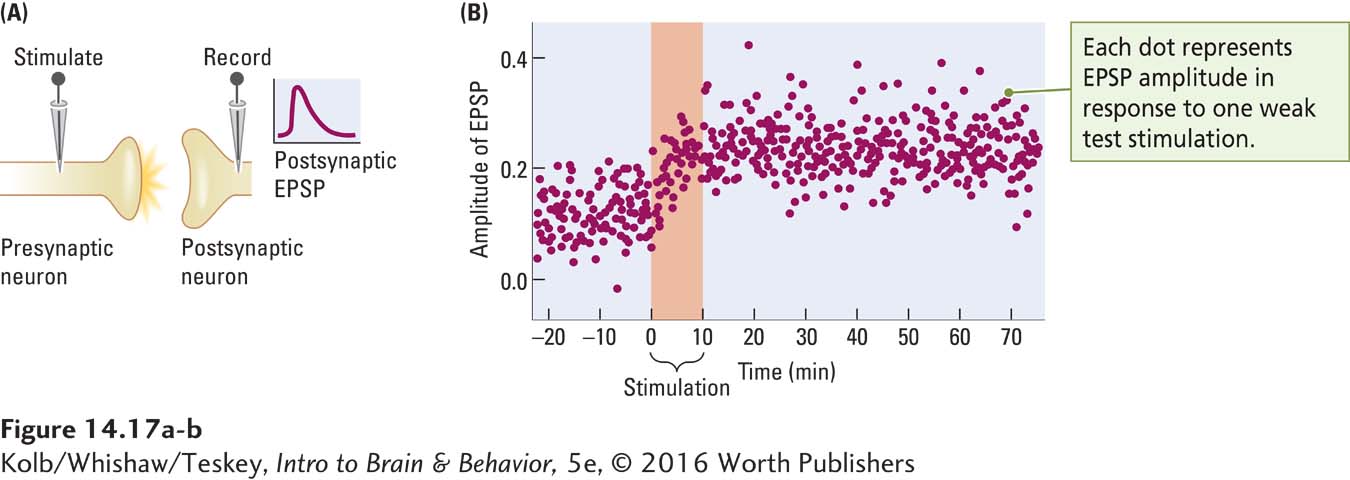
The discovery of LTP led to a revolution in thinking about how memories are stored. As investigators varied the stimulation that produced LTP, they discovered its opposite. Instead of using high-
If LTP and LTD form a basis for understanding synaptic changes underlying memory, two predictions follow. First, when animals learn problems, we should see enhanced LTP in the recruited pathways. Second, LTP should produce enduring changes in synaptic morphology that resemble those seen in memory. Both predictions appear to be true.
NMDA is shorthand for N-methyl-
The original studies of LTP concentrated on excitatory glutamate synapses. Glutamate is released from the presynaptic neuron and acts on two different types of receptors on the postsynaptic membrane, the NMDA and AMPA receptors, as shown in Figure 14-18A. AMPA receptors ordinarily mediate responses produced when glutamate is released from a presynaptic membrane. They allow sodium ions (Na+) to enter, depolarizing and thus exciting the postsynaptic membrane. The initial amplitude of the EPSP in Figure 14-17A is produced by this AMPA receptor action.
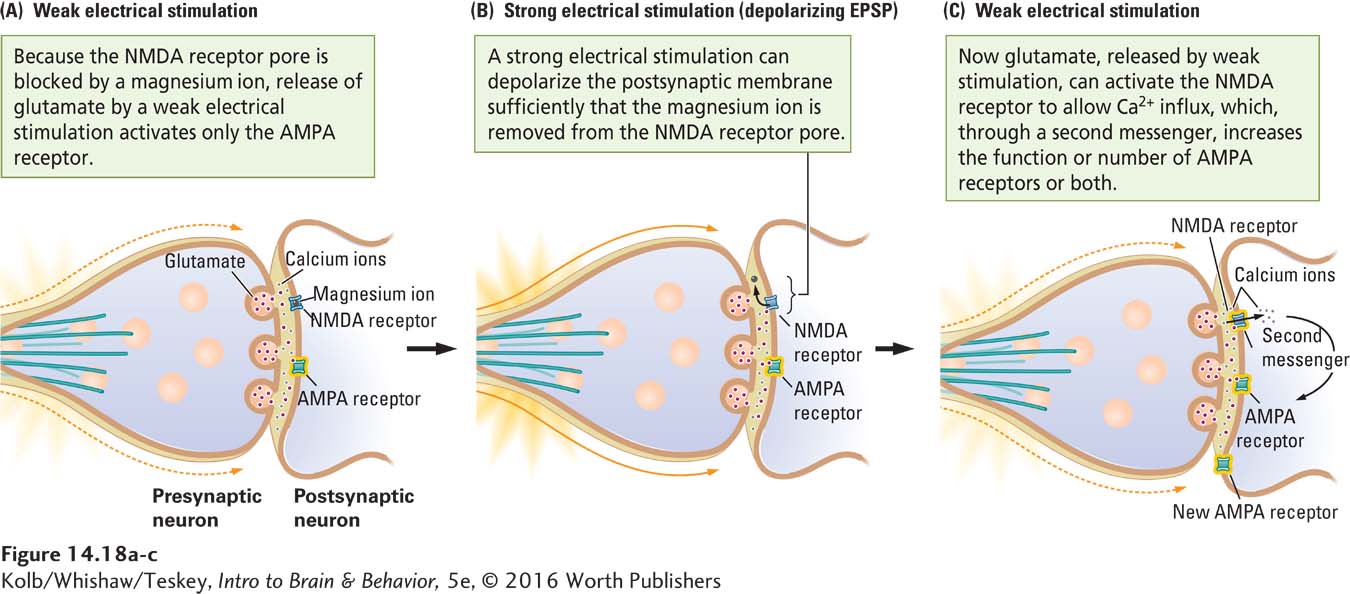
NMDA receptors do not usually respond to glutamate, because their pores are blocked by magnesium ions (Mg2+). NMDA receptors are doubly gated ion channels that can open to allow the passage of calcium ions if two events take place at approximately the same time:
- The postsynaptic membrane is depolarized, displacing the magnesium ion from the NMDA pore (Figure 14-18B). The strong electrical stimulation delivered by the experimenter serves as a way of displacing magnesium.
- NMDA receptors are activated by glutamate from the presynaptic membrane (Figure 14-18C).
With the doubly gated NMDA channels open, calcium ions enter the postsynaptic neuron and act through second messengers to initiate the cascade of events associated with LTP. These events include increased responsiveness of AMPA receptors to glutamate, formation of new AMPA receptors, and even retrograde messages to the presynaptic terminal to enhance glutamate release. One or more of these actions produce the final EPSP amplitude in Figure 14-17B.
Although the studies generated by the Bliss and Lømø discoveries have focused on excitatory synapses, experiments on inhibitory GABA interneurons demonstrate phenomena similar to LTP and LTD, labeled LTPi and LTDi.This discovery was a surprise. At the time it was generally believed that inhibitory neurons were not plastic, but they definitely are. It appears that plasticity of GABAergic (inhibitory) synapses plays some fundamental role in modulating networks of excitatory neurons.
Studying LTP mechanisms highlights neuroscientists’ uncertainty over where plastic changes are located. Our discussion emphasizes postsynaptic changes, but a strong case can be made that key presynaptic changes are at work too (e.g,, MacDougall & Fine, 2014). In general, plasticity likely requires change on both sides of the synapse. The presynaptic side, by virtue of being activated first, may prove key in the early phases of synaptic changes.
Measuring Synaptic Change
In principle, experience could change the brain in either of two ways: by modifying existing circuitry or by creating novel circuitry. In actuality, the plastic brain uses both strategies.
Modifying Existing Circuits
The simplest way to find synaptic change is to look for gross changes in the morphology of dendrites. Essentially, dendritic spines are extensions of the neuron membrane that allow more space for synapses. Cells that have few or no dendrites have limited space for inputs, whereas cells with complex dendritic protrusions may have space for tens of thousands of inputs.
Figure 3-5 shows how dendrites branch from three types of neurons.
So more dendrites mean more connections. Change in dendritic structure, therefore, implies change in synaptic organization. In complex neurons, such as pyramidal cells, 95 percent of synapses are on the dendrites. Measuring the extent of dendritic changes allows us to infer synaptic change.
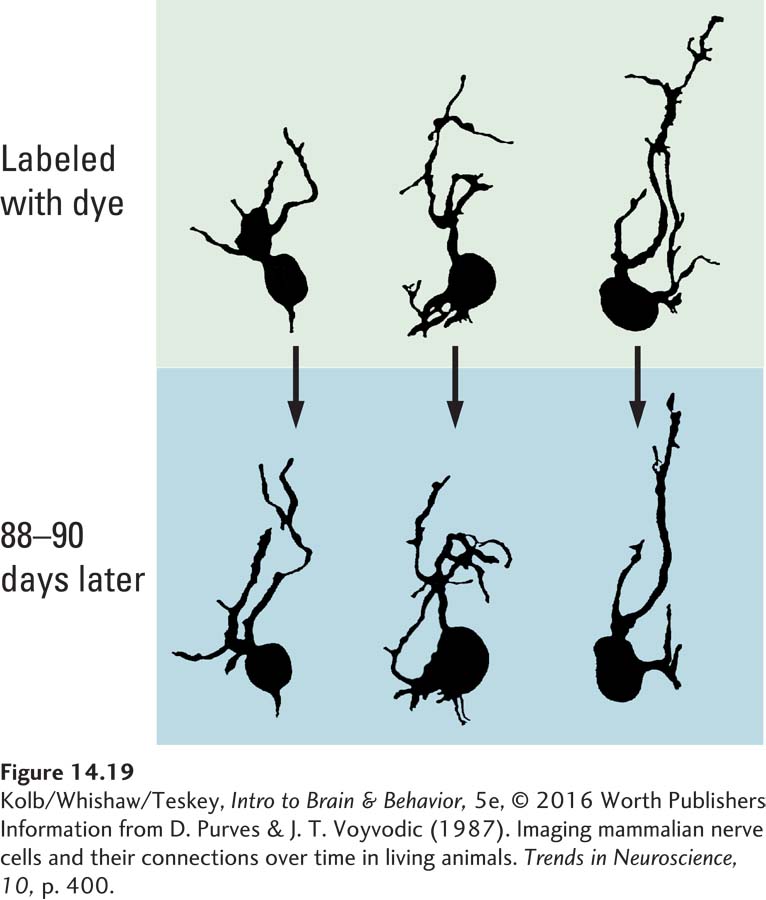
Dendritic shape is highly changeable. Dale Purves and his colleagues (Purves & Voyvodic, 1987) labeled cells in the dorsal root ganglia of living mice with a dye that allowed them to visualize the cells’ dendrites. When they examined the same cells at intervals ranging from a few days to weeks, they identified obvious qualitative changes in dendritic extent (Figure 14-19). We can assume that new dendritic branches have new synapses and that lost branches mean lost synapses.
Focus 5-5 diagrams how dendritic spines form and explains why they provide the structural basis for behavior.
An obvious lesson from the Purves studies is that neuronal morphology is not static: neurons change their structure in response to changing experiences. As they search for neural correlates of memory, researchers can take advantage of this changeability by studying variations in dendritic morphology that are correlated with specific experiences, such as learning some task.
What do changes in dendritic morphology reveal? Let us consider a given neuron that generates more synaptic space. The new synapses can provide either additional contacts between neurons that were already connected with that neuron or contacts between neurons not formerly connected. Figure 14-20 illustrates examples of these distinct synapse types.
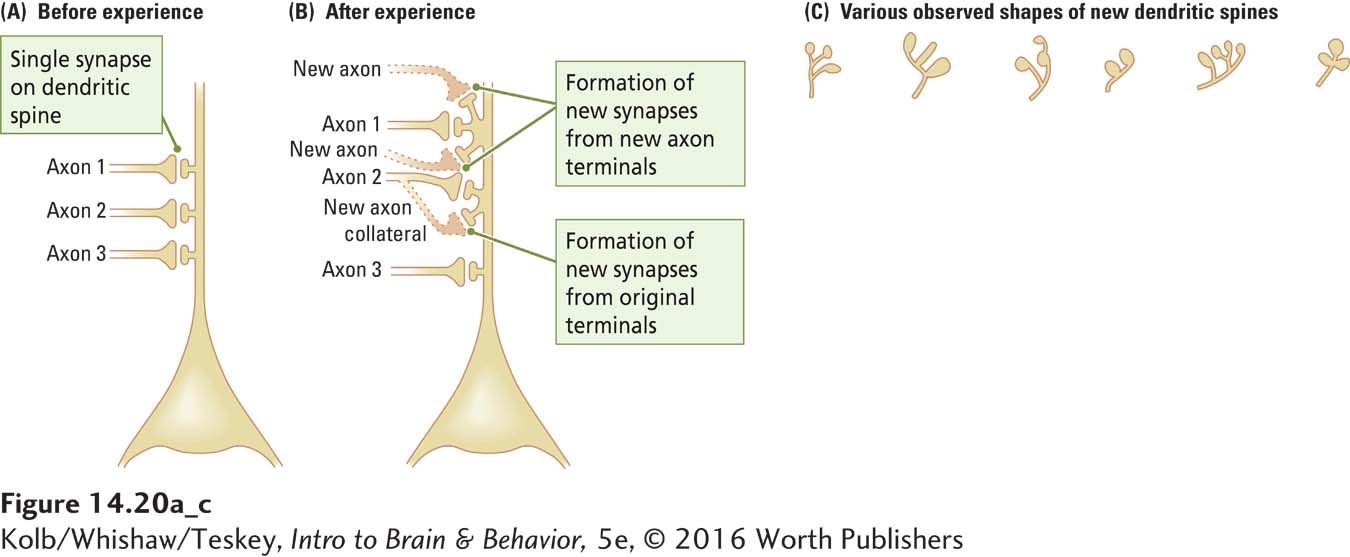
New synapses can result either from the growth of new axon terminals or from the formation of synapses along axons as they pass by dendrites (Figure 14-20A and B). In both cases, new synapses correspond to changes in local or regional circuitry rather than to the development of new connections between distant parts of the brain. Forming new connections between widely separated brain regions would be difficult in a fully grown brain: the dense plexus of cells, fibers, and blood vessels blocks the way.
Thus, the growth of new synapses indicates modifications to basic circuits already in the brain. This strategy has an important implication for the location of synaptic changes underlying memory. During development, the brain forms circuits to process sensory information and to produce movement (behavior). These are the circuits most likely to be modified to form memories (see Figure 14-5).
Creating Novel Circuits
Only 25 years ago, the general assumption was that the mammalian brain did not generate new neurons in adulthood. The unexpected discovery in the 1970s that the brains of songbirds such as canaries grow new neurons to produce songs in the mating season led researchers to reconsider this assumption. They found that the adult mammalian brain, too, is capable of generating new neurons.
This discovery emerged from directly injecting animals with a compound—
Experiment 7-1 confirms the hypothesis that hippocampal neurons contribute to memory formation.
The BrdU technique yielded considerable evidence that the mammalian brain, including the primate brain, can generate neurons destined for the olfactory bulb, the hippocampal formation, and possibly even the frontal and temporal lobe neocortex (Eriksson et al., 1998; Gould et al., 1999). The reason is not yet clear, but adult neurogenesis may enhance brain plasticity, particularly in processes underlying learning and memory. Elizabeth Gould and her colleagues (1999) showed, for example, that generation of new neurons in the hippocampus is enhanced when animals learn explicit memory tasks.
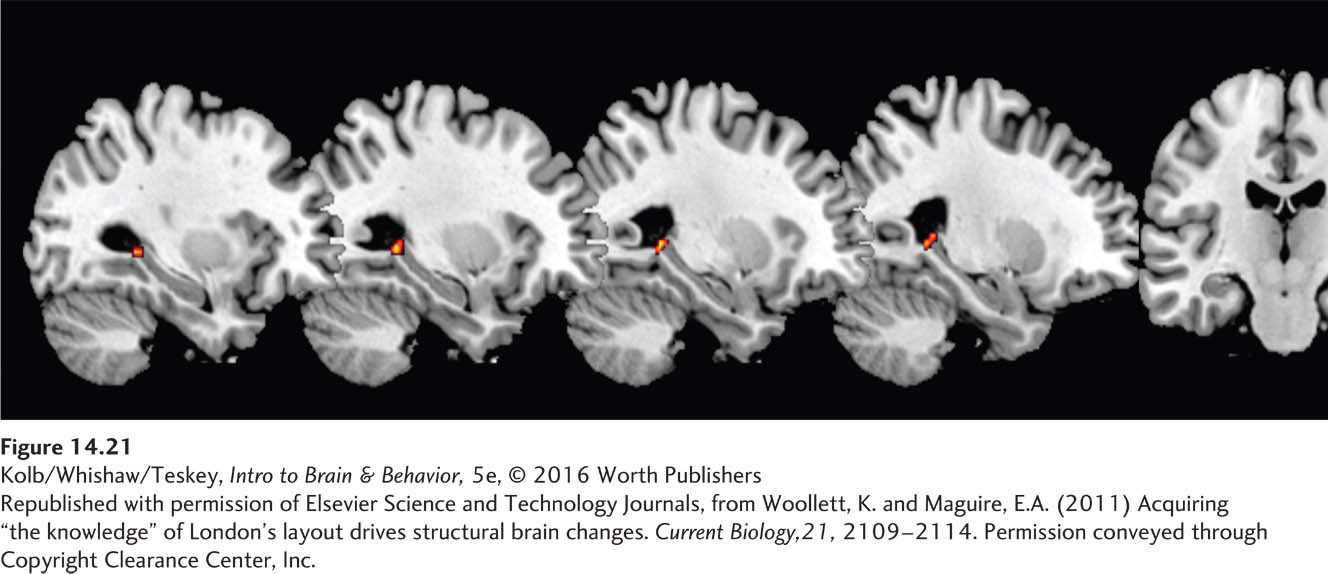
Experience appears to increase the generation of these new neurons. A fascinating demonstration of experience driving neurogenesis comes from a study by Katherine Woollett and Eleanor Maguire (2011). We noted in Section 14-3 that London taxi drivers, who must learn the locations of and pass an exam on central London’s roughly 25,000 irregular streets, have larger-
Woollett and Maguire recorded structural MRIs from the would-
Enriched Experience and Plasticity
Section 8-4 details Hebb’s first enrichment exercise—
One way to stimulate animals’ brains is to house the animals in environments that provide sensory or motor experience. Donald Hebb (1947) took laboratory rats home and gave them the run of his kitchen. After an interval, Hebb compared these “enriched” rats with a group that had remained caged in his laboratory at McGill University: he trained both groups to solve various mazes. The enriched animals performed better, and Hebb concluded that one effect of the enriched experience is to enhance later learning. This important conclusion laid the foundation for the U.S. Head Start programs, which provide academic experience for preschool-
Subsequent investigators have opted for a more constrained enriched enclosure. For example, in our own studies, we place groups of about six rats in enclosures. The enclosures give animals a rich social experience as well as extensive sensory and motor experience. The most obvious consequence is increased brain weight—
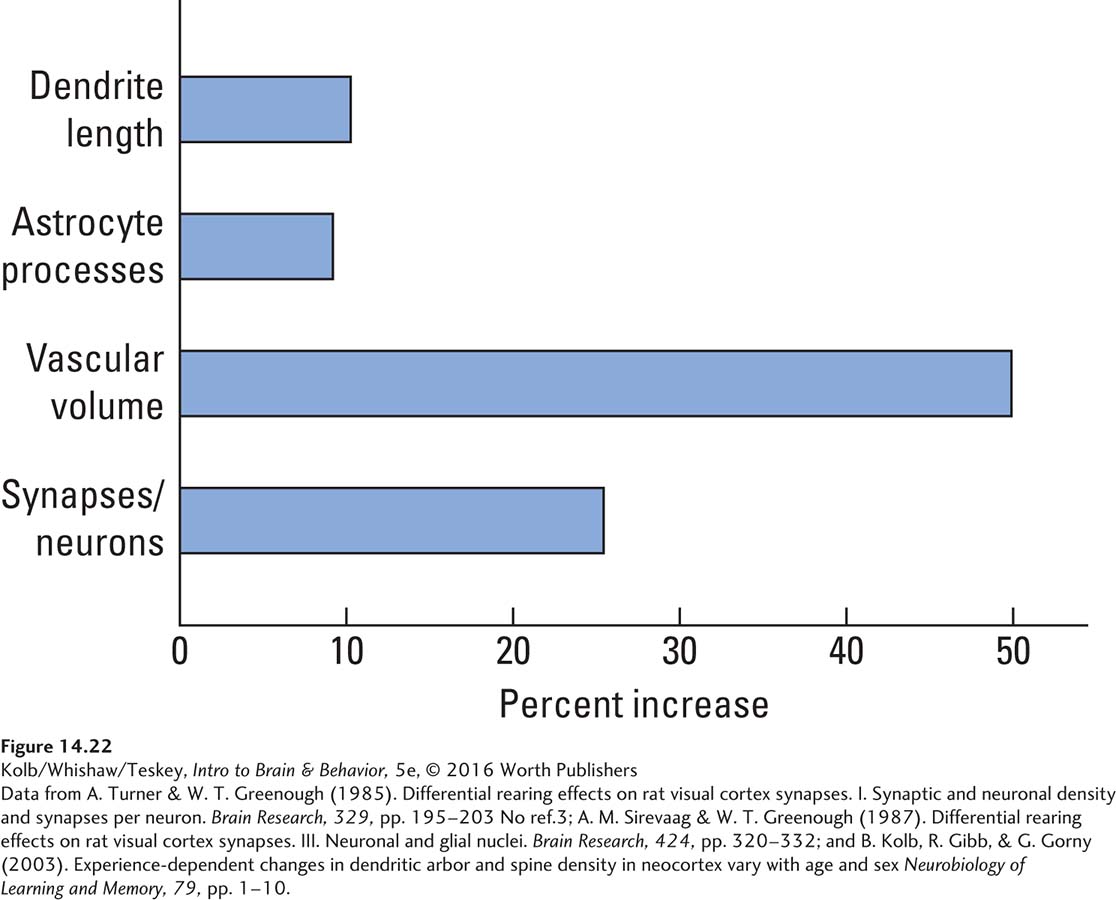
The key question is: What is responsible for the increased brain weight? A comprehensive series of studies by Anita Sirevaag and William Greenough (1988) used light-
Mitochondria power the cell. Figure 3-10 diagrams these organelles in the context of the neuron’s other internal components.
Animals with enriched experience have more synapses per neuron and also more astrocytes, more blood capillaries, and higher mitochondrial volumes. Clearly, when the brain changes in response to experience, the expected neural changes take place, and adjustments in the metabolic requirements of the now larger neurons take place as well.
Gerd Kempermann and his colleagues (1998) sought to determine whether experience actually alters the number of neurons in the brain. To test this idea, they compared neuronal generation in the hippocampi of mice housed in a complex environment with that of mice reared in a laboratory cage. They located the number of new neurons by injecting the animals with BrdU several times while they were living in complex housing.
The new neurons generated in the brain during the experiment incorporated the BrdU. When the researchers later looked at the hippocampi, they found more new neurons in the complex-
Sensory or Motor Training and Plasticity
Studies showing neuronal change in animals housed in a complex environment demonstrate that large areas of the brain can change with such experience. This finding leads us to ask whether specific experiences produce synaptic changes in localized cerebral regions. One way to approach this question is to give animals specific experiences, then see how their brain has changed. Another way is to look at the brain of people who have had a lifetime of some particular experience. We consider each research strategy separately.
Manipulating Experience Experimentally
In humans, only about half the optic fibers cross. Figure 9-10 diagrams the pathways.
Fen-
Chang and Greenough placed a patch over one eye of each rat, then trained the animals in a maze. The visual cortex of only one eye received input about the maze, but their two hemispheres’ auditory, olfactory, tactile, and motor regions were equally active as the animals explored. A comparison of the neurons in each hemisphere revealed that those in the visual cortex of the trained hemisphere had more extensive dendrites. Because the hemispheres did not differ in other respects, the researchers concluded that some feature associated with encoding, processing, or storage of visual input from training was responsible for forming new synapses.
Randy Nudo and his colleagues (1997) conducted complementary studies, mapping the motor cortex of monkeys. They noted striking individual differences in topography. The investigators speculated that individual variability might be due to each monkey’s experiences up to the time at which the cortical map was derived. To test this idea directly, Nudo and colleagues trained two groups of squirrel monkeys to retrieve banana-
EXPERIMENT
Question: Does the learning of a fine motor skill alter the cortical motor map?
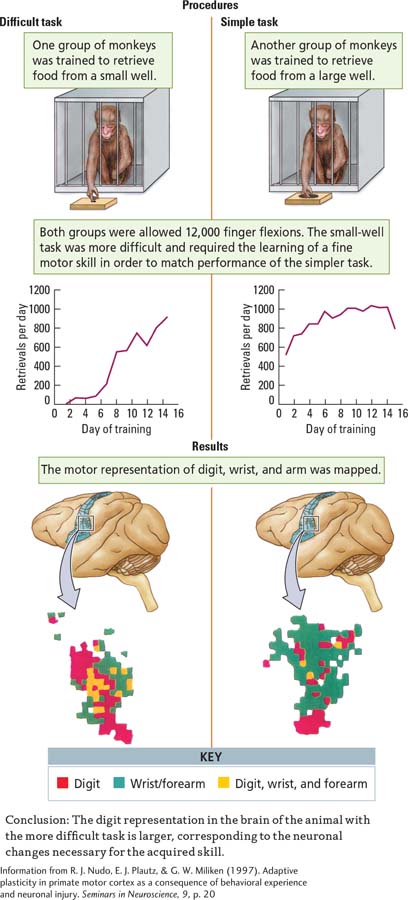
Monkeys in the two groups were matched for number of finger flexions, which totaled about 12,000 for the entire study. The monkeys trained on the small well improved with practice, making fewer finger flexions per food retrieval as training proceeded. Maps of forelimb movements, shown in the Results section, were produced by microelectrode stimulation of the cortex. The maps show systematic changes in the animals trained on the small but not on the large well. Presumably, these changes result from the more demanding motor requirements of the small-
Most studies demonstrating plasticity in the motor cortex have been performed with laboratory animals whose cortex was mapped by microelectrode stimulation. Today, imaging techniques such as transcranial magnetic stimulation (TMS) and functional magnetic resonance imaging (fMRI) make it possible to show parallel results in humans who have special motor skills. For example, right-
Thus, the functional organization of the motor cortex is altered by skilled use in humans. It can also be altered by chronic injury in humans and laboratory animals. Jon Kaas (2000) showed that when the sensory nerves in one limb are severed in monkeys, large-
Focus 11-5 recounts Ramachandran’s therapy for minimizing phantom limb pain.
Similar results can be found in the cortical maps of people whose limb has been amputated. For example, Vilayanur Ramachandran (1993) found that when a hand amputee’s face is brushed lightly with a cotton swab, the person has a sensation of the amputated hand being touched. Figure 14-23 illustrates the rough map of the hand that Ramachandran was actually able to chart on the face. The likely explanation is that the face area in the motor cortex has expanded to occupy the deafferented limb cortex, but the brain circuitry still responds to the cortical activity as representing input from the limb. This response may explain the phantom limb pain often experienced by amputees.
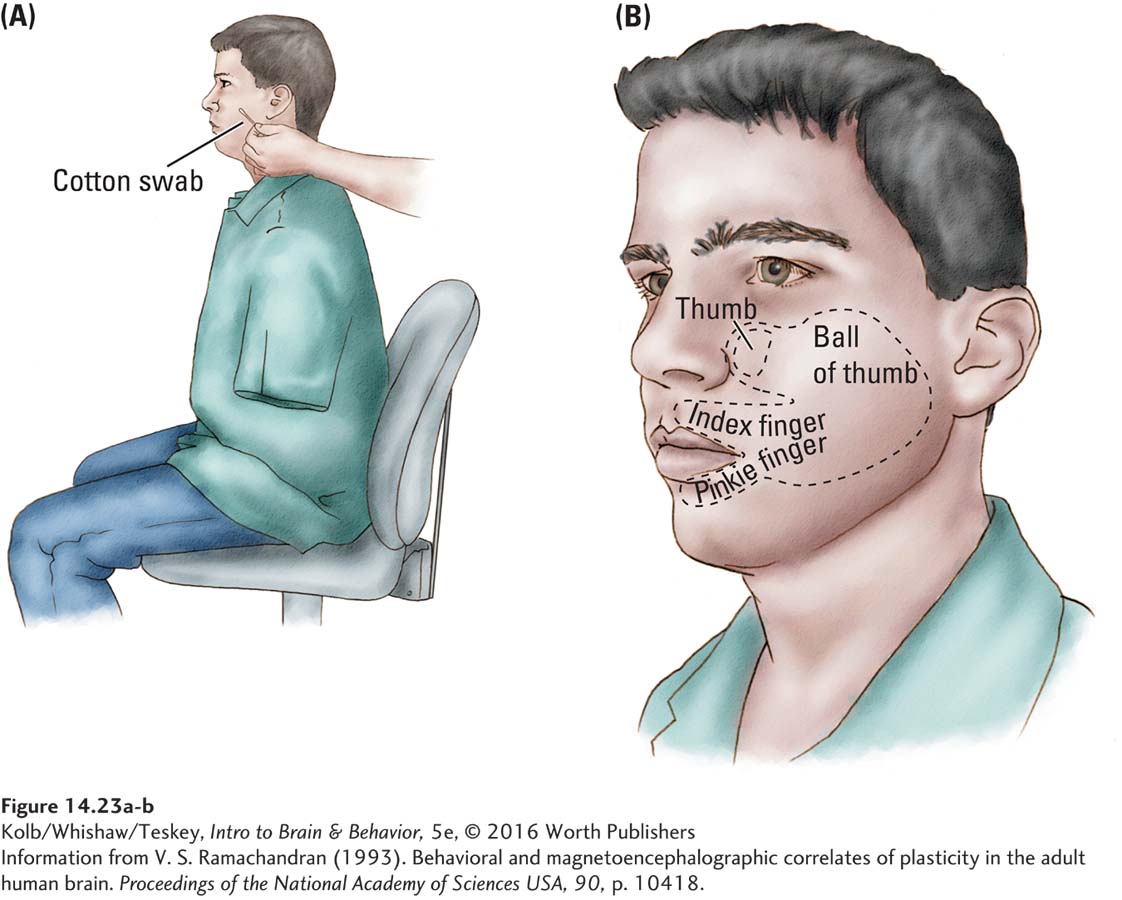
The idea that experience can alter cortical maps can be demonstrated with other experiences. For example, if animals are trained to make certain digit movements over and over again, the cortical representation of those digits expands at the expense of the remaining motor areas. Similarly, if animals are trained extensively to discriminate among different sensory stimuli such as tones, the auditory cortical areas responding to those stimuli increase in size.
Sections 10-4 and 15-4 discuss music’s benefits for the brain.
As described in Research Focus 14-5, Movement, Learning, and Neuroplasticity, one effect of musical training is to alter the motor representations of the digits used to play different instruments. We can speculate that musical training probably alters the auditory representations of specific sound frequencies as well. Both changes are essentially forms of memory, and the underlying synaptic changes likely take place on the appropriate sensory or motor cortical maps.
14-5
Movement, Learning, and Neuroplasticity
Many lines of research show that practicing a motor skill—
Presumably, musical skill improves with practice, but are other abilities enhanced too? Patrick Ragert and colleagues (2003) showed that professional pianists not only have better motor skills in their fingers but enhanced somatosensory perception as well.
When the researchers measured the ability to detect subtle sensory stimulation of the fingertips, they found that the pianists were more sensitive than controls. They also found that the enhanced tactile sensitivity was related to the hours per day that the musicians spent practicing.
The investigators then asked whether the enhanced perceptual ability precluded further improvement in the musicians. Surprisingly, when both the musicians and controls were given a 3-
This result implies that well-
Not all motor learning is good, however. Many musicians develop focal hand dystonia—
Typically, dystonia afflicts musicians who practice trying to make perfect finger movements on their instruments. Musicians at high risk include string players, who receive vibratory stimulation at their fingertips. The constant practice has been suggested to lead not only to improved musical ability but also to distorted or disordered cortical motor maps. Synchronous activation of the digits by the vibration leads to this unwanted side effect.
Victor Candia and colleagues (2003) reasoned that musicians’ dystonia was probably an example of disordered learning and could be treated by retuning the motor map. The investigators used magnetoencephalography (MEG) to measure changes in sensory-
At the beginning of the study, the musicians with dystonia had a disordered motor map: the finger areas overlapped one another. In training, each subject used a hand splint tailored to his or her hand. The splint allowed for immobilizing different fingers while the subjects independently moved the others.
After 8 days of training for about 2 hours per day, the subjects showed marked alleviation in the dystonic symptoms, and the neuroimaging showed a normalization of the cortical map, with distinct finger areas. Thus, training reversed the learned changes in the motor map and treated the dystonia. The musicians had actually learned a disorder, and they were able to unlearn it.

Experience-Dependent Change in the Human Brain
According to Ramachandran’s amputee study, the human brain appears to change with altered experience. But this study did not examine neuronal change directly; it inferred neuronal change from behavior. The only way to examine synaptic change directly is to look directly at brain tissue. In living humans, this is not an option, but the brain of people who died of something other than neurological causes can be examined and the structure of their cortical neurons related to their experiences.
Wernicke’s area contributes to speech and to language comprehension; see Figure 10-18.
One way to approach this idea is to look for a relation between neuronal structure and education. Arnold Scheibel and his colleagues conducted many such studies in the 1990s (e.g., Jacobs and Scheibel, 1993; Jacobs, Scholl, and Scheibel, 1993). In one, they found a relation between dendrite size in Wernicke’s area and level of education. In the brain of deceased people with a college education, the cortical neurons from this language area had more dendritic branches than did those from people with a high-
Figure 15-16 diagrams tasks that consistently show, on average, that females’ verbal fluency surpasses males’ and that males outperform females on spatial reasoning tasks.
Another way to look at the relation between neurons in Wernicke’s area and behavior is to take advantage of the now well-
Finally, these investigators took a slightly different approach to the link between experience and neuronal morphology. They began with two hypotheses. First, they suggested a relation between the complexity of dendritic branching and the nature of the computational tasks performed by a brain area.
To test this hypothesis, they examined the dendritic structure of neurons in different cortical regions that handle different computational tasks. For example, when they compared the structure of neurons corresponding to the somatosensory representation of the trunk with those for the fingers, they found the latter to have more complex cells (Figure 14-24). They reasoned that the somatosensory inputs from receptive fields on the chest wall would constitute less of a computational challenge to cortical neurons than would those from the fingers and that the neurons representing the chest would therefore be less complex.
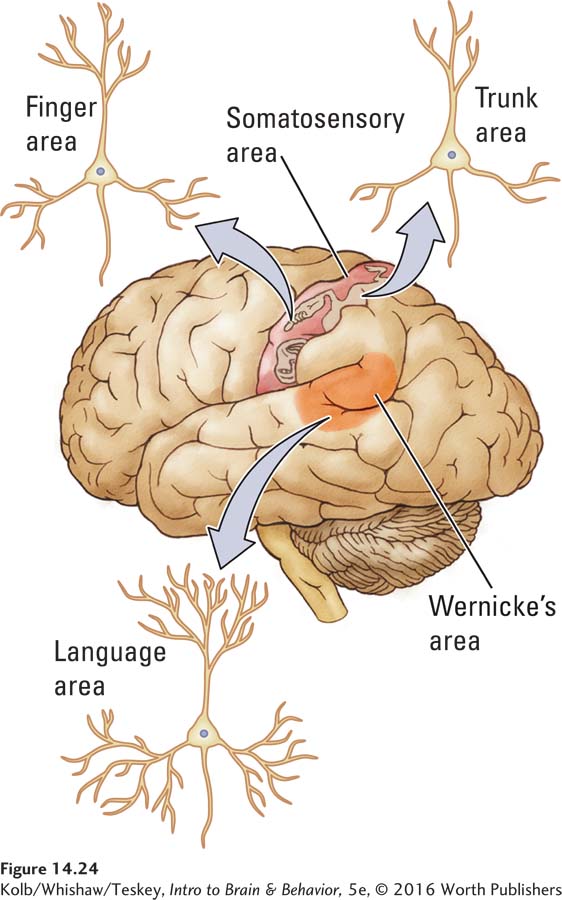
The group’s second hypothesis was that dendritic branching in all regions is subject to experience-
For example, especially large differences in trunk and finger neurons appeared in the brain of people who had a high level of finger dexterity maintained over long periods (say, career word processors). In contrast, no difference between trunk and finger neurons was found in sales representatives. Remember, Scheibel and colleagues conducted their research before portable electronic devices entered the workplace. We would not expect a good deal of specialized finger use among sales reps of that time and thus less complex demands on their finger neurons.
In summary, although the studies showing a relation between experience and neuronal structure in humans depend on correlations rather than actual experiments, the findings are consistent with those observed in experimental studies of other species. We are thus led to the general conclusion that specific experiences can produce localized changes in the synaptic organization of the brain and that such changes form the structural basis of memory.
Epigenetics of Memory
An enigma in the search for neural mechanisms underlying memory is the fact that whereas memories remain stable over time, all cells are constantly undergoing molecular turnover. The simplest explanation for this is epigenetic: specific sites in the DNA of neurons involved in specific memories might exist in either a methylated or a nonmethylated state.
Figure 3-25 illustrates two aspects of methylation: histone and DNA modification.
Courtney Miller and colleagues (2010) tested this idea directly by measuring methylation in the hippocampi of rats that underwent contextual fear conditioning (see Experiment 14-1). They showed that fear conditioning is associated with rapid methylation, but if they blocked methylation, there was no memory. The investigators conclude that epigenetic mechanisms mediate synaptic plasticity broadly, but especially in learning and memory. One implication of these results is that cognitive disorders, including memory defects, could result from aberrant epigenetic modifications (for a review, see Day et al., 2015).
Plasticity, Hormones, Trophic Factors, and Drugs
The news media often report that psychoactive drugs can damage your brain. Some drugs certainly do act as toxins and can selectively kill brain regions, but a more realistic mode of drug action is to change the brain. Although not many studies have looked at drug-
Hormones and Plasticity
Section 6-5 explains the classes, functions, and control that hormones exert; Section 8-4, their organizing effects during development; Section 12-5, their activating effects in adulthood.
Levels of circulating hormones are critical both in determining brain structure and in eliciting certain behaviors in adulthood. Although the structural effects of hormones were once believed to be expressed only in the course of development, current belief is that adult neurons also can respond to hormonal manipulations with dramatic structural changes. We consider the actions of gonadal hormones and stress hormones here.
Research findings have established that structural differences in cortical neurons of male and female rats depend on gonadal hormones. More surprising, perhaps, is that gonadal hormones continue to influence cell structure and behavior in adulthood. Elizabeth Hampson and Doreen Kimura (1988) showed that women’s performance on various cognitive tasks changes throughout the menstrual cycle as their estrogen levels fluctuate.
Changes in estrogen level appear to alter the structure of neurons and astrocytes in the neocortex and hippocampus, which probably accounts for at least part of the performance fluctuation. Figure 14-25 illustrates changes in dendritic spines in the hippocampal cells of female rats at different phases of their 4-
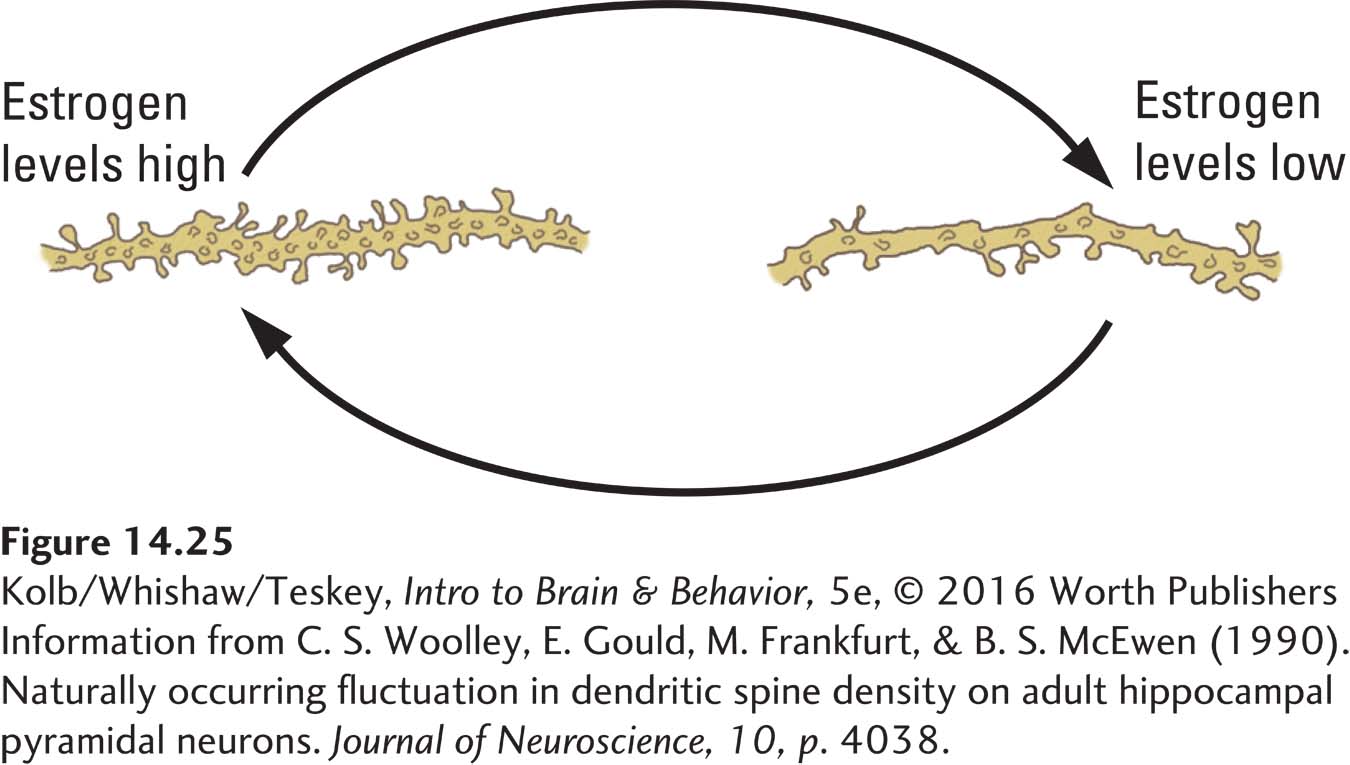
Curiously, estrogen’s influence on cell structure may differ in the hippocampus and neocortex. Jane Stewart found, for example, that when the ovaries of middle-
This question is also relevant to middle-
When the body is stressed, the pituitary gland produces adrenocorticotrophic hormone (ACTH), which stimulates the adrenal cortex to produce steroid hormones, the glucocorticoids. Important in protein and carbohydrate metabolism, controlling sugar levels in the blood, and the absorption of sugar by cells, glucocorticoids have many actions on the body, including the brain. Robert Sapolsky (1992) proposed that glucocorticoids can sometimes be neurotoxic.
Figure 6-19 illustrates the body’s stress response.
In particular, he found that with prolonged stress, glucocorticoids appear to kill hippocampal cells. Elizabeth Gould and her colleagues (1998) showed that even brief periods of stress can reduce the number of new granule cells produced in the hippocampi of monkeys, presumably through the actions of stress hormones. Evidence of neuron death and reduced neuron generation in the hippocampus has obvious implications for animal behavior, especially for processes such as spatial memory. Finally, Richelle Mychasiuk and her colleagues (2015) showed that stress has contrasting epigenetic effects in the hippocampus and prefrontal cortex, with virtually no overlap between males and females.
In sum, hormones can alter the brain’s synaptic organization and even the number of neurons in the brain. Little is known today about the behavioral consequences of such changes. It is likely that hormones can alter the course of plastic changes in the brain, possibly through epigenetic mechanisms.
Neurotrophic Factors and Plasticity
Section 8-2 explains how neurotrophic factors send these signals.
Neurotrophic factors, chemical compounds listed in Table 14-2 that signal stem cells to develop into neurons or glia, also act to reorganize neural circuits. The first, nerve growth factor (NGF), was discovered in the peripheral nervous system more than a generation ago. NGF is trophic (nourishing) in the sense that it stimulates neurons to grow dendrites and synapses and in some cases promotes neuronal survival.
A Hebb synapse—
Trophic factors produced in the brain by neurons and glia can affect neurons both through cell membrane receptors and by actually entering the neuron to act internally on its operation. Trophic factors may be released postsynaptically, for example, to act as signals that can influence the presynaptic cell. Experience stimulates their production, so neurotrophic factors have been proposed as agents of synaptic change. For example, brain-
Although many researchers would like to conclude that BDNF has a role in learning, this conclusion does not necessarily follow. When animals solve mazes, their behavior differs from their behavior when they remain in cages. So we must first demonstrate that changes in BDNF, NGF, or any trophic factor are actually related to forming new synapses. Nevertheless, if we assume that trophic factors do act as synaptic change agents, then we should be able to use increased trophic factor activity during learning as a marker for where to look for changed synapses associated with learning and memory.
Psychoactive Drugs and Plasticity
Many people regularly use the stimulant caffeine, and some use more stimulating psychoactive drugs such as nicotine, amphetamine, or cocaine. The long-
One experimental demonstration of these changes is drug-
The sea slug Aplysia becomes more sensitive to a stimulus after repeated exposure. Psychoactive drugs appear to have a parallel action: they lead to increased behavioral sensitivity to their actions. For example, a rat given a small dose of amphetamine may show increased activity. When the rat is given the same dose of amphetamine on subsequent occasions, the increase in activity is progressively larger. If no drug is given for weeks or even months and then amphetamine is given in the same dose as before, behavioral sensitization picks up where it left off and continues to progress. Some long-
The parallel between drug-
In a series of studies, Terry Robinson and his colleagues found dramatic increases in dendritic growth and spine density in rats sensitized to amphetamine, cocaine, or nicotine relative to rats that received injections of a saline solution (Robinson & Kolb, 2004). Experiment 14-3 compares the effects of amphetamine and saline treatments on cells in the nucleus accumbens in the basal ganglia. Neurons in amphetamine-
EXPERIMENT
Question: What effect do repeated doses of amphetamine, a psychomotor stimulant, have on neurons?
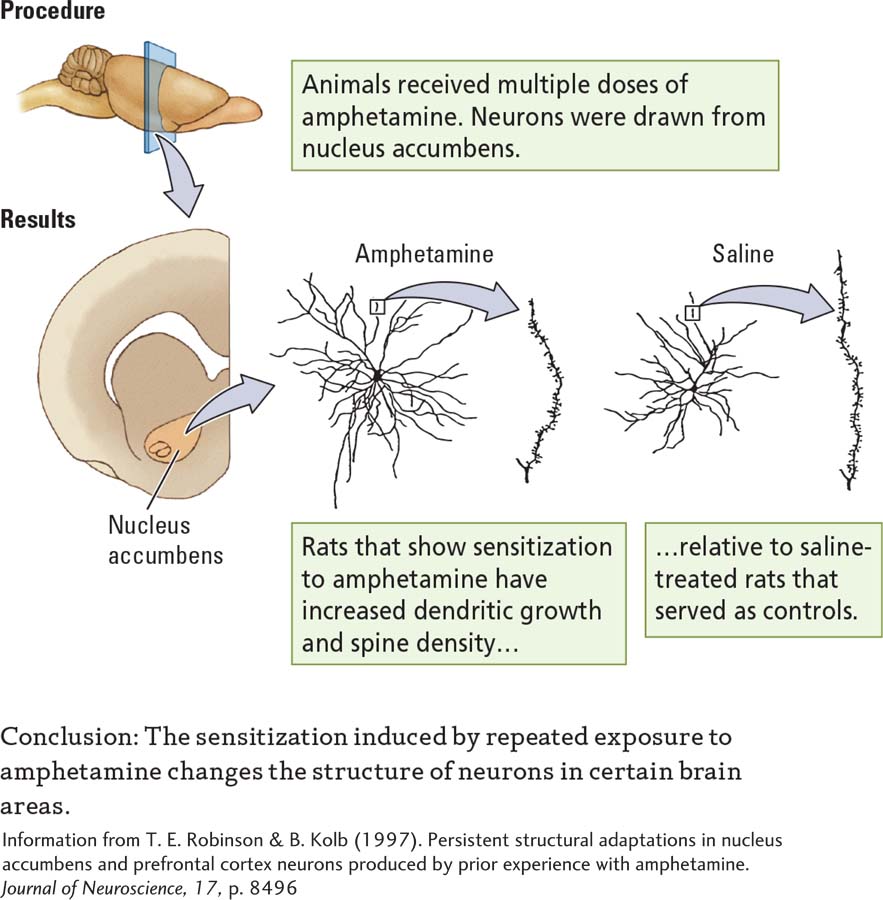
These plastic changes were not found throughout the brain. Rather, they were localized to regions such as the prefrontal cortex and nucleus accumbens that receive a large dopamine projection. Dopamine is believed to factor significantly in the rewarding properties of drugs (Wise, 2004). Other psychoactive drugs also appear to alter neuronal structure. Marijuana, morphine, and certain antidepressants change dendritic length and spine density, although in ways different from those of stimulants. Morphine, for example, reduces dendritic length and spine density in the nucleus accumbens and prefrontal cortex (Robinson & Kolb, 2004).
What do drug-
Figure 12-30 shows enhanced connectivity in prefrontal and limbic circuits in nicotine-
Why prior drug exposure has this effect is as yet unknown, but obviously drug taking can have long-
Some Guiding Principles of Brain Plasticity
Brain plasticity will continue as a fundamental concept underlying research into brain–
1. Behavioral Change Reflects Brain Change
The brain’s primary function is to produce behavior, but behavior is not static. We learn and remember, we think new thoughts or visualize new images, and we change throughout life. All these processes require changes in neural networks. Whenever neural networks change, behavior, including mental behavior, also changes. A corollary is especially important as neuroscientists search for treatments for brain injuries or behavioral disorders: To change behavior, we must change the brain.
2. All Nervous Systems Are Plastic in the Same General Way
Investigators study neuroplasticity in species ranging from worms and insects to fish, birds, and mammals.
Even the simplest animals, such as the roundworm C. elegans, can show simple learning that correlates with neuronal plasticity. The molecular details may differ between simple and complex systems, but the principles of neuroplasticity appear to be conserved across both simple and complex animals. This conservation allows more studies of neural plasticity among a wider range of animal species than in most areas of neuroscience.
3. Plastic Changes Are Age-
Section 8-4 traces how brain organization details change rapidly—
The brain responds to the same experiences differently at different ages—
4. Prenatal Events Can Influence Brain Plasticity Throughout Life
Section 8-4 recounts benefits of tactile stimulation; Focus 8-2, epigenetic factors in the autism spectrum; Focus 6-2, the tragedy of fetal alcohol spectrum disorder.
Prenatal experiences can alter brain organization. Potentially negative experiences, such as prenatal exposure to recreational or prescription drugs, and positive experiences, such as tactile stimulation of the mother’s skin, may alter gene expression or induce other epigenetic effects that produce enduring effects on brain organization. Even paternal or maternal experiences before conception can alter later offsprings’ brain development and organization.
5. Plastic Changes Are Brain Region Dependent
Figure 12-19 diagrams these prefrontal regions.
Although we are tempted to expect plastic changes in neuronal networks to be fairly general, it is becoming clear that many experience-
6. Experience-
Metaplasticity is a property of a lifetime’s interaction among different plastic changes in the brain. As an animal travels through life, infinite experiences can alter its brain organization. A lifetime’s experiences might interact. Housing animals in complex environments produces profound changes in their neural network organization, but prior exposure to psychoactive drugs completely blocks the enrichment effect. Conversely, although complex housing does not block drug effects, enrichment markedly attenuates them. Prenatal events can affect later drug effects: prenatal tactile stimulation of the mother, for example, reduces the later effects of psychoactive drugs on the child.
7. Plasticity Has Pros and Cons
We have mainly emphasized the neuroplastic changes that can support improved motor and cognitive function. But as noted for the effects of psychoactive drugs, plastic changes in neural networks can also interfere with behavior. Drug addicts whose prefrontal cortex has been altered are prone to poor judgment in their personal life. People who have posttraumatic stress disorder show altered blood flow in the amygdala and cingulate cortex. That’s the bad news.
Encouraging plastic changes that reverse these prefrontal alterations is the good news, and it is associated with a loss of the prefrontal disorder. Age-
14-4 REVIEW
Structural Basis of Brain Plasticity
Before you continue, check your understanding.
Question 1
Repeated high-
Question 2
LTPi and LTDi are found in ____________ neurons.
Question 3
Structural changes underlying memory include changes in both ____________ and ____________.
Question 4
Learning complex spatial information has been linked to increased gray matter in the ____________.
Question 5
The progressive increase in behavioral actions in response to repeated administration of a drug is called ____________.
Question 6
How can plastic changes in the brain produce adverse effects?
Answers appear in the Self Test section of the book.